EMA set to simplify biosimilar approval
The EMA has asked the industry to share their thoughts on its draft guidelines for developing and assessing biosimilars. The idea is to make it easier for patients in the EU to get access to biosimilars, while also keeping Europe a good choice for developers.
The new approach for equivalence testing of biosimilars, which is outlined in a new draft reflection paper, is designed to reduce the amount of clinical data required for the development and approval of biosimilar medicines. It builds on the 2024 Procedure for European Union guidelines and related documents within the pharmaceutical legislative framework‘. The EMA‘s consultation will end at the end of September and will be implemtented in 2026.
While Europe has long been dominated by homegrown biosimilar developers, foreign competition is gaining ground. Between 2022 and 2024, 11% of EMA-approved biosimilars came from India or South Korea (CGAR 30%), a 3% increase over the past three years. This trend is expected to accelerate as more non-European firms enter the space.
Biosimilars have been authorised based on studies comparing them to their reference medicine. This includes a comparability exercise on quality aspects of the active substances of the biosimilar and the reference medicine, and the demonstration of clinical efficacy and safety of the biosimilar in confirmatory clinical trials.
Building on two decades experience with biosimilar medicines and advances in analytical methods, the EMA’s draft Scientific guidelines’. reflection paper now suggests that demonstrated structural and functional comparability, together with comparative data on pharmacokinetics may be sufficient to demonstrate similarity with the reference medicine. If adopted, the new rules are set to reduce the need for extensive clinical efficacy studies. Waiving certain clinical data requirements would simplify the development and evaluation process while maintaining the highest standards of safety and efficacy. Between now and 2030, 69 biologics in Europe and 118 in the U.S. will lose exclusivity, creating an opportunity eight times larger than the 2012–2014 period.
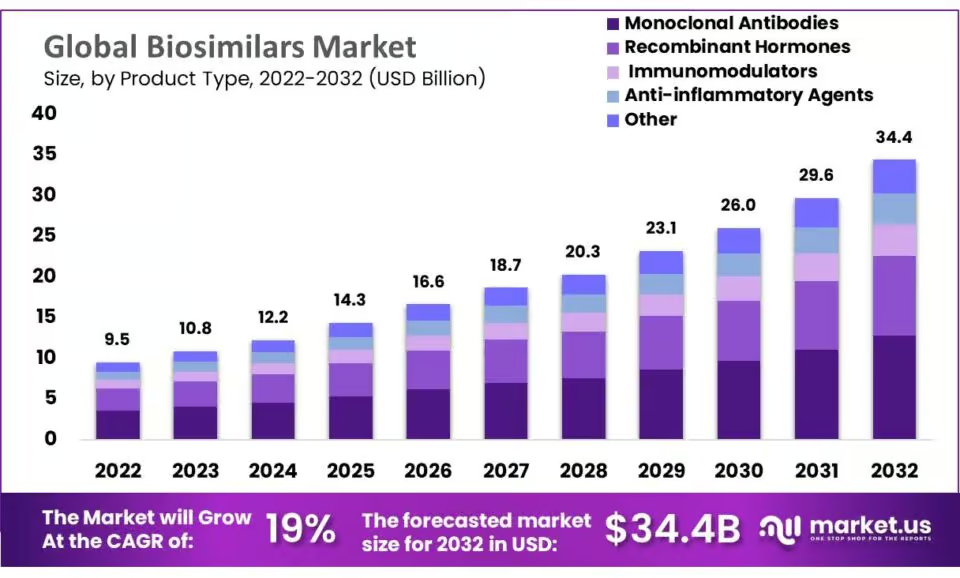
Biosimilars have become important therapeutic options improving patient access to essential treatments such as costly biologics. By providing competition in the market, they can expand patient access to critical medicines. Today, biosimilars are available in six major therapeutic areas, contributing to 6.9 billion patient treatment days since the first EU approvals in 2006. However, more than 90 % of biosimilar development is focused on oncology and immunology, leaving other therapeutic areas underserved.
The EMA’s more streamlined approach would ultimately ensure wider availability of biosimilar medicines to patients in the EU.




 FDA
FDA