Scarlet Therapeutics has raised £3.2 million (€3.7 million) to take therapies based on red blood cells (RBCs) to in vivo proof of concept, building on evidence that natural and lab-grown cells have comparable half-lives.
ADVERTISEMENT
Bayer AG is strengthening its ophthalmology portfolio with the acquisition of Perfuse Therapeutics for up to $2.45bn. The focus is on a Phase II candidate for glaucoma and diabetic retinopathy with potential as a disease-modifying therapy.
If one follows the figures in the Swiss Biotech Report 2026, the green lights are more prominent than the red warning signals. The Swiss biotech industry continued its growth momentum in 2025 and even reached new record highs. According to the industry report presented in early May at Swiss Biotech Day in Basel, total revenue among companies classified as biotech — which in this case explicitly does not include Roche and Novartis, for example — rose to CHF 7.5 billion, up from CHF 7.2 billion the previous year. The drivers were a growing number of market-ready products and persistently strong demand for specialized CDMO services.
In the evolving landscape of drug discovery and new design, scientists and pharmaceutical innovators continually strive to develop therapies that are both highly selective and clinically effective, while addressing targets previously deemed “undruggable”. In recent years, macrocycles – a class of large, ring-shaped molecules – have emerged as a compelling solution at the crossroads between traditional small molecules and large biologics, offering a blend of high specificity, rich chemical diversity and promising pharmacological profiles.
Basel-based Windward Bio did it again and has raised another three-digit millions – $165 million to be exact – to advance a pipeline of long-acting immunology therapies—much of it sourced through in-licensing deals with Chinese partners. Its lead candidate, WIN378, is moving into Phase III, with first clinical readouts expected from 2026.
Cytospire Therapeutics has raised a £61 million (€71 million) Series A round, positioning the British biotech to advance pan-gamma-delta T-cell engagers (TCEs) into cancer clinical trials.
Belgian biopharma UCB has moved quickly from testing the waters in autoimmune T-cell engagers to making one of the largest bets yet on the modality, agreeing to acquire San Diego-based Candid Therapeutics for up to US$2.2bn.
Can in‑space manufacturing help cancer patients take complex medicines at home? For a technology that aims to do just that, UK‑based BioOrbit has just raised £9.8m (around €11.3m) – the “world’s largest” seed round of for in‑space manufacturing.
Valneva SE, the French specialty‑vaccine developer, has completed an €84m reserved offering, adding a useful cash buffer just as the group enters a high‑risk window for its Lyme disease and shigellosis‑vaccine candidates.
With the launch of Servier Ventures announced in January and a €200 million commitment to biotech investment, Servier is stepping up its engagement in early-stage innovation in oncology and neurology. European Biotechnology Magazine spoke with Alexis Vandier, Global Head of Servier Ventures, about the fund’s strategy, investment focus, and Europe’s role in the global biotech ecosystem.



 Bayer
Bayer
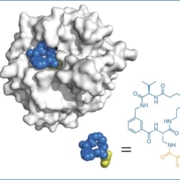 Sevan Habeshian & Christian Heinis et al. https://www.nature.com/articles/s41467-022-31428-
Sevan Habeshian & Christian Heinis et al. https://www.nature.com/articles/s41467-022-31428- Getty Images, for Unsplash+
Getty Images, for Unsplash+

 Planet Volumes via Unsplash+
Planet Volumes via Unsplash+ Valneva
Valneva Servier
Servier