The French biopharmaceutical company, PLL Therapeutics, announced positive results from a phase I/II clinical trial for its investigational therapy PLL001 to treat amyotrophic lateral sclerosis (ALS), a progressive neurodegenerative disease that causes degeneration of motor neurons and loss of muscle control.
ADVERTISEMENT
Tag Archive for: Neurology
Newron Pharmaceuticals has secured up to €38mn to advance Phase III trials of evenamide, a drug that, if successful, could represent one of the first circuit-modulating therapies in schizophrenia.
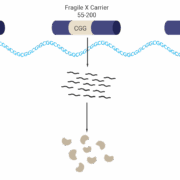
In a US$450m deal with UK-based Kaerus Bioscience Ltd, Servier SA has acquired the development and commercialisation rights to KER-0193, a Phase II-ready drug candidate for the treatment of rare Fragile X syndrome, the most common monogenic cause of autism.
French targeted DNA gene therapy specialist EG427 has closed a €27m Series B financing co-led by VC investor Andera Partners and BPI France to initiate Phase I testing of a Herpes Simplex Virus 1 (nrHSV-1) vector that specifically targets type C neurons to suppress dysfunctions of the bladder in people with spinal injury.
Nxera Pharma Ltd has got a Phase II milestone from US licencor Neurocrine Biosciences for its first-in class oral schizophrenia candidate NBI-1117568.
Evotec SE has reached a predefined milestone worth €20m in the CRO’s partnership with Bristol Myers Squibb (BMS).
ALS specialist NeuroSense Therapeutics Ltd announces it will be collaborating with Swiss Lonza AG to identify exosome-based biomarkers to advance theranostics in neurodegeneration.
Spanish Ferrer Internacional, SA has aquired the ex-US rights to VRG50635 from Verge Genomics Inc to commercialise the oral Phase I experimental ALS treatment.
University of Sheffield spinout gets £5m for gene therapies targeting ALS and FTD, advancing towards clinical trials.
uniQure N.V. is to sell a portion of its royalty rights from the net sales o its hemophilia B gene therapy etranacogene dezaparvovec-drbl to HealthCare Royalty and Sagard Healthcare for a gross purchase price of up to $400m in cash. This financing provides uniQure with immediate, non-equity-dilutive capital to continue to invest in our AAV gene therapy pipeline and platform and to advance potentially breakthrough gene therapies for patients, including AMT-130 in Huntington’s disease, AMT-260 for refractory temporal lobe epilepsy, AMT-162 for SOD-1 ALS and other product candidates, statedMatt Kapusta, CEO of uniQure.
Etranacogene dezaparvovec-drbl is a gene therapy that reduces the rate of abnormal bleeding in eligible people with hemophilia B by enabling the body to continuously produce factor IX, the deficient protein in hemophilia B. It uses AAV5, a non-infectious viral vector, called an adeno-associated virus (AAV). The AAV5 vector carries the Padua gene variant of Factor IX (FIX-Padua) to the target cells in the liver, generating factor IX proteins that are 5x-8x more active than normal. These genetic instructions remain in the target cells, but generally do not become a part of a person’s own DNA. Once delivered, the new genetic instructions allow the cellular machinery to produce stable levels of factor IX.
Under the terms of the agreement, uniQure will receive an upfront cash payment of $375m in exchange for the lowest royalty tier on CSL Behring’s worldwide net sales of etranacogene dezaparvovec-drbl up to 1.85 times the purchase price until June 30, 2032 or, if such cap is not met by June 30, 2032, up to 2.25x the purchase price through December 31, 2038.
Amsterdam-based uniQure NV led the multi-year development of etranacogene dezaparvovec-drbl and completed an exclusive global license and commercialization agreement with CSL Behring in May 2021. Etranacogene dezaparvovec-drbl HEis approved for the treatment of hemophilia B in the US, Europe and the United Kingdom. CSL Behring also is pursuing registration in additional countries.
uniQure expects the transaction will extend its cash runway into the second quarter of 2026, assuming receipt of the contractual milestone payment for the first sale of etranacogene dezaparvovec-drbl in the US.



 FRAXA Ewsearch Organisation
FRAXA Ewsearch Organisation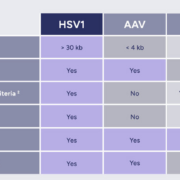 EG 427
EG 427 Nxtera Pharma Ltd
Nxtera Pharma Ltd