Ipsen has announced the immediate withdrawal of its cancer drug Tazverik across all indications after serious safety issues emerged in an ongoing Phase III trial.
ADVERTISEMENT
Tag Archive for: Rare disease
argenx delivered two significant updates on the same day: positive Phase 3 results in ocular myasthenia gravis (oMG) and its first full year of operating profitability. Together, the announcements show a company expanding its lead medicine into a new patient population while doing so from a position of financial strength.
The European Commission has approved Amgen’s Uplizna in generalized myasthenia gravis, moving the big biotech into a market served by argenx, Johnson & Johnson and UCB.
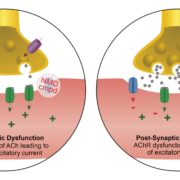
Researchers at NDM Pharma A/S and Aarhus University have demonstrated that their oral CIC-1 blocker NMD670 restores crosstalk between neurons and muscles in Myasthenia gravis.
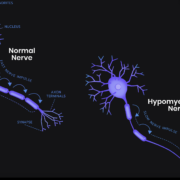
Rare disease specialist SynaptixBio Ltd has got its second FDA orphan drug designation for a subtype of TUBB4A leukodystrophy.


 Unsplash+
Unsplash+