The Basel region is one of the world’s leading life-science clusters. Pharmaceutical companies, biotech firms and research institutions benefit from a unique combination of talent, infrastructure and international collaboration. As the industry continues to grow, the demand for scalable laboratory and office environments within this innovation ecosystem is increasing.
ADVERTISEMENT
Europe continues to play a pivotal role in the global biologics landscape, combining scientific excellence, regulatory maturity and a strong industrial foundation.
The AI Circular Economy Conference 2026, organised by nova-Institute, brought together 116 participants from 15 countries in Cologne and online to explore the transformation of the chemical and materials industry supported and accelerated by artificial intelligence.
Research and innovation are considered key resources for European competitiveness – and essential location factors for research-intensive industries such as the Life Sciences. The European Union is increasingly responding to this finding with a multifaceted catalogue of regulations and measures: alongside horizontal and sector-specific legal frameworks, data-law infrastructures as well as operational initiatives are being introduced, which are intended to accelerate procedures and facilitate the translation of research into healthcare delivery and into the market.
3PBIOVIAN consolidates its position as a trusted partner for the development and manufacturing of complex biological products, combining scientific excellence, regulatory expertise, and strong social commitment.
Customized Enzymes and Reagents from New England Biolabs. Creating the right partnership is essential when pioneering a new life science product.
In an interview with European Biotechnology Magazine during J.P. Morgan Week in San Francisco, Dr. Jian Zhang, Acting CEO of BioDlink, explains how a partnership-first approach and deep ADC expertise help him offer biotech companies a competitive edge.
As the regulatory strategy becomes a boardroom-level concern for biotech executives pursuing global expansion, knowing how the Saudi Food and Drug Authority (SFDA) and the European Medicines Agency (EMA) differ is crucial, particularly as Saudi Arabia accelerates its ambition to become a global life sciences hub. Although both regulators adhere to international norms, their differences in review speed, procedural flexibility, and strategic positioning can have a direct impact on competitive advantage, investment planning, and time to market.
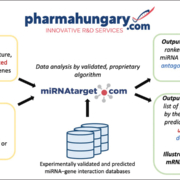
Pharmahungary Group (PH) developed miRNAtarget, a miRNA discovery software tool based on miRNA-mRNA molecular networks. PH holds patent families on 4 cardio-protective microRNAs families (protectomiRs) and seeks for co-development partners and/or early stage investors. PH also offers its miRNA discovery and development service platform that includes efficacy testing from in vitro to large animal models in various therapeutic areas.
This article examines the critical success factors for PEGylated therapies and the decision-making process for when a custom PEG may be warranted rather than an off-the-shelf solution. The essential role of a contract development and manufacturing organization (CDMO) with deep expertise in activated PEG chemistry is highlighted.
Authors: Raymond Behrendt, Silvia Kaden-Vagt, Lotfi Bouchekioua, Merck KGaA, Darmstadt, Germany


 SONUVO
SONUVO 3PBiovian
3PBiovian



 BioDlink
BioDlink
 Pharmahungary Group
Pharmahungary Group Merck KGaA, Darmstadt Germany
Merck KGaA, Darmstadt Germany