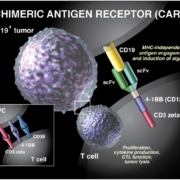
Swiss Novartis AG has received US market authorisation for its first-in-class CAR-T cell therapy Kymriah (tisagenlecleucel-T). As severe cytokine storms seemed to be generally linked to CAR-T therapies such as Kymriah, the FDA also authorised Roche’s anti-IL-6R antibody Actemra (tocilizumab) as first-in-class treatment to manage them.
ADVERTISEMENT
The US Food and Drug Administration has granted US market authorisation to Boehringer Ingelheim’s biosimilar anti-TNFa-antibody adalimumab.
Supported by the European Fund for Strategic Investments (EFSI), the European Investment Bank (EIB) has granted a €25m loan to the Austrian pediatric cancer specialist Apeiron Biologics AG to boost its pipeline of antibodies, fusion proteins and low molecular weight checkpoint inhibitors.
The possibilities offered by CRISPR/Cas9, TALENs and other new genome editing technologies are making the mouths of plant breeders water – and not just because the methods give seed developers new options when it comes to optimising traits such as yield, resistance to environmental stress, diseases or pests. Gene-edited plants could soon make inroads even with Europe’s strict regulators, and throw open the door to tastier, healthier food.
In mid-June, the German Federal Constitutional Court asked the German President not to sign legislation designed to implement the EU Unified Patent Court (UPC, EU Agreement 16351/12) and Unitary Patent (EU Regulation 1257/12). Now, there are hints for a further delay. The constitutional complaint behind the delay, rumoured to be brought up by a German life sciences IP expert, has led to a stop of hiring German judges for the UPC.
Danish Genmab A/S’ anti-CD38 antibody daratumumab has met the endpoint of progression-free survival in an interim analysis of Genmab’s pivotal Phase III study aimed at receiving FDA approval as first-line therapy in multiple myeloma.
Merck Ventures has joined the Series A investor syndicate of the British immuno-oncology specialist Macrophage Pharma Ltd.
Scientists at Swedish Karolinska Institute have identified a factor that supports growth of midbrain dopaminergic (mDA) neurons, the nerve cells that degenerate during of Parkinson’s disease.
Evotec’s immunology spin-out Topas Therapeutics has inked a multi-year R&D collaboration with pharma major Eli Lilly. The initial focus of research, which will be financed by Eli Lilly, is on identification of antigens that specifically induce a T regulatory cell response in inflammatory and auto-immune disorders.
Swedish specialty pharmaceutical company Pharmalink AB Stockholm has Fredrik Johansson as new Chief Financial Officer.