In the evolving landscape of drug discovery and new design, scientists and pharmaceutical innovators continually strive to develop therapies that are both highly selective and clinically effective, while addressing targets previously deemed “undruggable”. In recent years, macrocycles – a class of large, ring-shaped molecules – have emerged as a compelling solution at the crossroads between traditional small molecules and large biologics, offering a blend of high specificity, rich chemical diversity and promising pharmacological profiles.
ADVERTISEMENT
With the launch of Servier Ventures announced in January and a €200 million commitment to biotech investment, Servier is stepping up its engagement in early-stage innovation in oncology and neurology. European Biotechnology Magazine spoke with Alexis Vandier, Global Head of Servier Ventures, about the fund’s strategy, investment focus, and Europe’s role in the global biotech ecosystem.
The 2026 Professor Wallhäußer Innovation Award was presented during this year’s PharmaCongress & PharmaTechnica 2026 at the end of March. First place went to AstraZeneca for the project “Automated Reading of Agar Plates for Environmental Monitoring with AI”.
According to the WHO, one adult in six globally is affected by infertility, but beyond this statistic, infertility is best understood as a couple’s problem, even when the underlying biology sits with one partner. The way we currently handle infertility issues is more about bypassing biology through procedure, with in vitro fertilisation (IVF) as its backbone. But while IVF is indispensable, success rates still vary, pushing biotech to step in and find new solutions.
For decades, biotech companies were formed around a discovery: a promising biological signal, a novel target, a platform emerging from academic research. Now a different formation model is gaining momentum: venture studios. These entities don’t just fund startups, they assemble them, testing hypotheses, building teams and infrastructure, and only then spinning out companies designed to scale.
Twelve emerging life science startups to showcase enabling technologies at the SLAS European Conference and Exhibition in Vienna, 19-21 May 2026
For years, Portugal sat just outside Europe’s main biotech conversation: scientifically credible, strong in research, but too small, too fragmented and too thinly financed to compete with the established hubs in Switzerland, the UK, France, Germany or the Nordics. That view is becoming harder to defend. Portugal still does not have the scale of Europe’s top biotech markets, but it is building something more durable than a collection of isolated startups.
Ten young European biotechs are heading into 2026 with the kind of momentum that can quickly turn promising science into defining data: first clinical entries, platform-to-pipeline transitions, and funding rounds large enough to accelerate execution. Some are pushing new modalities into hard disease areas, while others are compressing discovery timelines and expanding what’s druggable. What they all share is a clear momentum that makes them especially worth following this year.
As advanced therapeutic medicinal products (ATMPs) and other next-generation biologics move toward approval, Chemistry, Manufacturing and Controls (CMC) continues to be the most frequent source of regulatory concern. This article highlights recurring CMC barriers and best practices to mitigate approval delays or refusals.
The Unified Patent Court and the unitary patent were established to make patent litigation in Europe easier, faster and more affordable. It has been particularly emphasized that the new system was designed to enable small and medium-sized enterprises to enforce their rights more effectively and rapidly. The reality now suggests differently. Litigation in Europe has become quite fragmented, more complex and more expensive than ever – a nice playground for deep pockets.


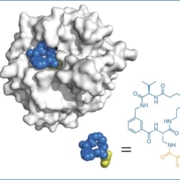 Sevan Habeshian & Christian Heinis et al. https://www.nature.com/articles/s41467-022-31428-
Sevan Habeshian & Christian Heinis et al. https://www.nature.com/articles/s41467-022-31428- Servier
Servier CONCEPT HEIDELBERG GmbH
CONCEPT HEIDELBERG GmbH Svitlana Hulko-freepik.com
Svitlana Hulko-freepik.com www.istockphoto.com/Yuri
www.istockphoto.com/Yuri SLAS
SLAS VanessaSpencer - stock.adobe.com
VanessaSpencer - stock.adobe.com Kelli McClintock auf Unsplash
Kelli McClintock auf Unsplash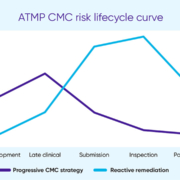 Cencora
Cencora Boehmert & Boehmert
Boehmert & Boehmert