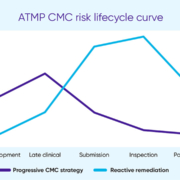
As advanced therapeutic medicinal products (ATMPs) and other next-generation biologics move toward approval, Chemistry, Manufacturing and Controls (CMC) continues to be the most frequent source of regulatory concern. This article highlights recurring CMC barriers and best practices to mitigate approval delays or refusals.
ADVERTISEMENT


 Cencora
Cencora