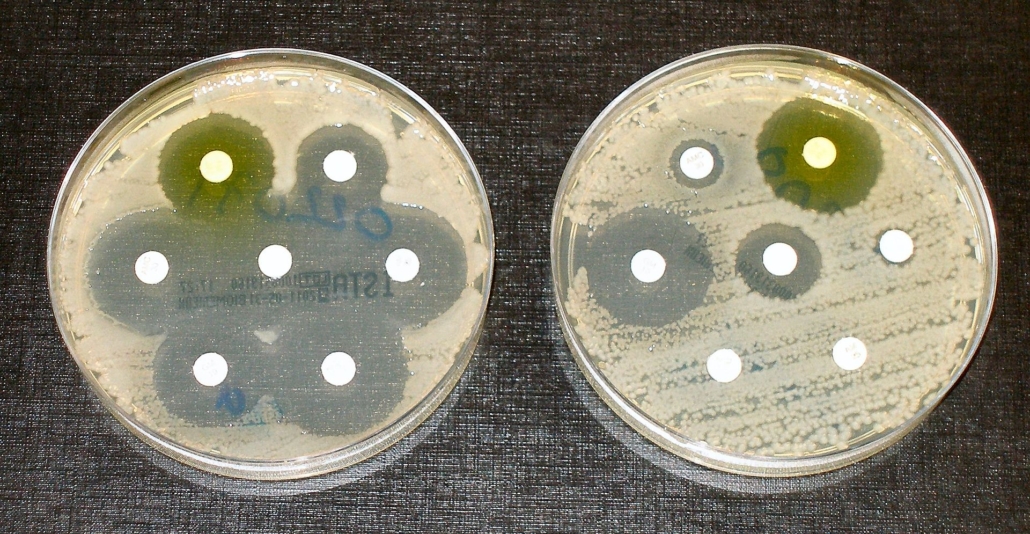
EU with poor success in fighting superbugs
The EU has made poor progress combatting antimicrobial resistance, a report from the European Court of Auditors (ECA) warns. Innovators need to be supported, urges the BEAM Alliance.
Although some progress has been made through EU action, there is little evidence so far to suggest that the health burden of antimicrobial resistance has been reduced, suggested the auditors at the beginning of the world antibiotics awareness week. On the one hand, the EU support has been useful in strengthening the Member States’ one-health approach to human, animal and environmental health to antibiotic resistance, particularly when it comes to better informed decisions and facilitated cooperation and exchange of experience between Member States. On the other hand, despite several candidates in the clinical pipeline, no breakthrough has been achieved in the development of new classes of antibiotics, the ECA claimed. According to the auditors, the failure of the market and the consequent inhibition of private sector research to combat antimicrobial resistance has not been sufficiently addressed by concrete EU initiatives.
The auditors recommend to improve support for the Member States, promote the prudent use of antimicrobial veterinary medicinal products and improve surveillance of antimicrobial resistance, i.e. by funding of the ECDC, and boost research into antibiotic resistance in the EU.
Currently, 90% of innovations in the AMR field come from SMEs. The developers’ industry association BEAM Alliance published a position paper underlining the need to rapidly provide market-based solutions supporting SME-driven innovation against antimicrobial resistance.
Albeit awareness to AMR has been raised at high political level, solutions are still slow to emerge, says the position paper. The situation is worsened by inadequate prescription practices and commercialisation constraints. Stakeholders at the national and global level must rapidly increase their efforts to design and implement a path forward to back the SMR innovators financially. Though the EIB is expected to raise €500m of loans next year as part of an €1bn AMR fund, currently there is no value-based reimbursement for novel compounds. Thus, several pull measures have to be implemented swiftly to prevent financial problems of innovators.
In its position paper, the Beam Alliance demands to align antimicrobial value and pricing with the true societal benefit; enable appropriate reimbursement paired with appropriate antimicrobial use, guarantee a minimal market footprint at product launch; and foster financial contributions from medical areas benefitting from antimicrobial use.
Today’s modern medicine would in large parts not be possible without working antibiotics, said Marc Gitzinger, Vice-President of the BEAM Alliance and CEO of Bioversys AG. Antibiotic resistance is a formidable threat to the achievements in healthcare of the last century. After most large pharma companies have abandoned the field, SMEs drive the innovation and have adequate answers to win the fight with novel therapies overcoming antibiotic resistance. However, investment into the field must be reinvigorated by immediate action from political leaders ensuring adequate pricing for life saving and appropriately used drugs, he stressed.



 CDC/Janice Haney Carr
CDC/Janice Haney Carr 3PBIOVIAN
3PBIOVIAN