High blood pressure reprograms immune system
Interferon-gamma overexpression leads to hypertension-induced damage to the blood-brain barrier by reprogrammed macrophages.
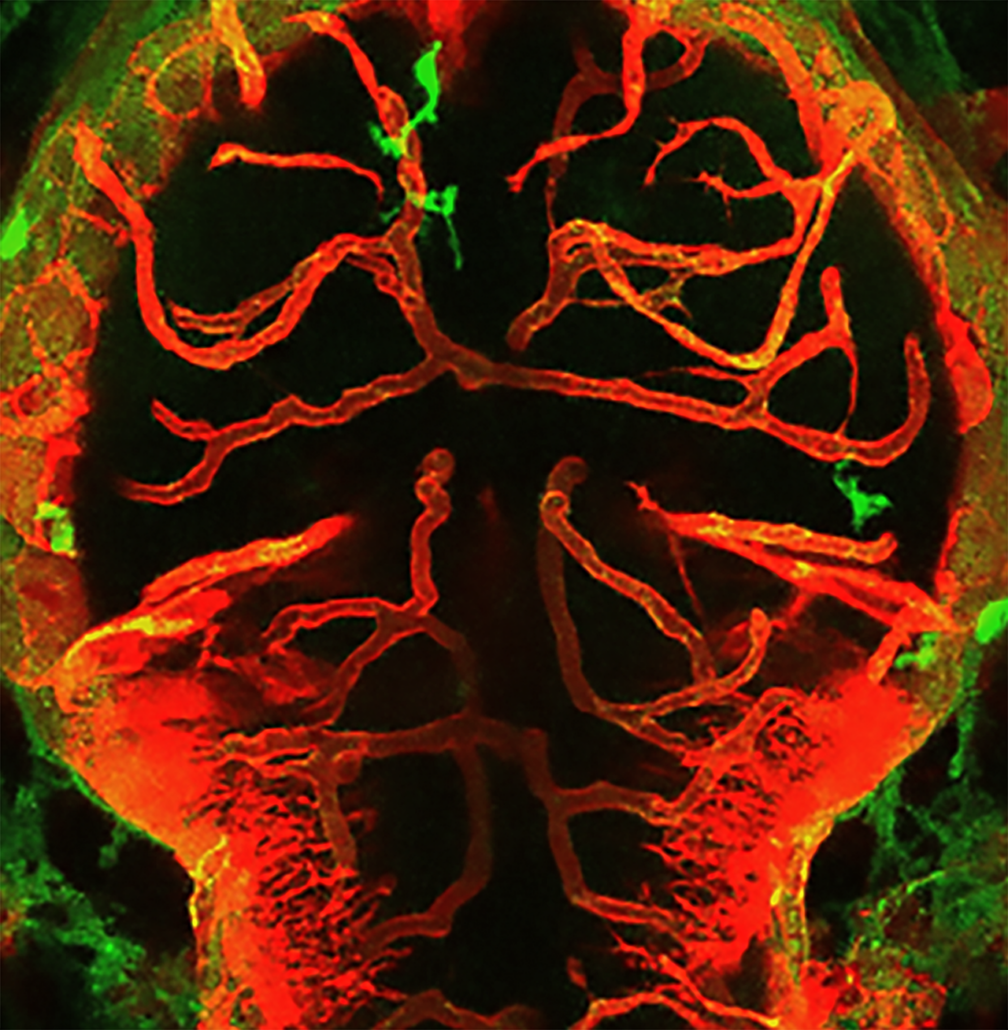
German scientists headed by Suphansa Sawamiphak from MDC Berlin have discovered in a zebrafish and mouse model of salt-induced hypertension that high blood pressure reprograms macrophages and microglial cells to attack the blood-brain barrier and damage blood vessels in the brain.
A key role in the endothelium-mediated damage to blood vessels by the brain’s macrophages and microglial cells, which are produced in greater numbers as a result of high blood pressure, is played by the messenger substance interferon-gamma (IFN-gamma), the researchers report in Cardiovascular Research. The weakening of the vascular walls and the blood-brain barrier induced by IFN-gamma overexpression induced the degradation of blood vessels in the brain, organ damage and cognitive deficits.
When first author Dilem Apaydin et al. genetically blocked the expression of the IFN-gamma receptor in the animal models, no organ damage occurred. In the zebrafish model, hypertension left no damage in blood vessels and blood-brain barrier. In mice, too, the blockade prevented typical side effects such as damage to the blood-brain barrier, degradation of blood vessels in the brain and cognitive deficits.
“Our results open up a completely new perspective on the role of inflammatory processes in the development of hypertension,” Sawamiphak emphasises. Now it is necessary to characterise the immune cells and immune modulators involved in more detail and to examine their role in higher animals up to humans. Should this be confirmed, there would be a new potential drug target that could benefit treatment-refractory patients in particular.


 Picture by Joanna Kosinska via Unsplash
Picture by Joanna Kosinska via Unsplash Sitryx Therapeutics
Sitryx Therapeutics SANOFI
SANOFI