Danish Novo Nordisk A/S has made an unexpected takeover bid for US biotech Metsera, Inc. This challenges the already agreed acquisition by Pfizer Inc. Novo offers US$6.5bn in cash, plus up to US$2.5bn in milestone payments. The total potential value is around US$9bn. Pfizer previously offered US$4.9bn plus US$2.4bn in milestones.
ADVERTISEMENT
The NATO is clearly priorising biotech to a defence priority with an annual budget of potentially up to €500m for biotech start-ups starting from 2026. At the opening of the first ever NATO Biotechnology Conference, NATO Secretary General Mark Rutte said: “Between last year and this year, we funded a total of 28 promising biotechnology companies through the Defence Innovation Accelerator for the North Atlantic (DIANA)”. Rutte indicated that DIANA’s budget could grow significantly in the medium term, potentially up to five-fold.
British GlaxoSmithKline plc (GSK) has entered a US$745m licensing agreement with Empirico Inc for EMP-012, a first-in-class siRNA candidate for COPD. EMP-012 targets the underserved non-type 2 inflammation market segment valued at US$600–700bn.
In a major bet on RNA medicine, Swiss Novartis AG announced to buy Avidity Biosciences Inc for US$12bn. Avidity’s Oligonucleotide Conjugate (AOC) platform allows Transferin receptor-targeted RNA delivery to muscle. With the acquisition Novartis secures access to three late-stage RNA drug candidates for muscular dystrophies.
GHO Capital has closed its fourth fund, GHO Capital IV LP, raising over €2.5bn (~US$2.9bn) to invest in global healthcare. The fund brings the firm’s total assets under management to around €9bn, making it Europe’s largest healthcare-focused private equity firm.
Hemab Therapeutics A/S has completed an oversubscribed US$157m Series C financing round led by Sofinnova Partners to accelarate and expand its pipeline in bleeding disorders and thrombosis treatments.
The French AI-synbio specialist Abolis Biotechnologies SAS has entered a strategic €140m alliance with API manufacturer EuroAPI SA under the IPCEI Med4Cure funding programme. Within the collaboration, Abolis is developing production chassis for microbial corticosteroid manufacturing. The goal: to make Europe independent from imports from Asia.
VC investments in biotech rose 70.9% in Q3 2025 to US$3.1bn, driven by Series D growth, M&A activity, and Fed interest rate cuts, reports Global Data. J.P. Morgan reports same trends but different figures.
California-based biotech Electra Therapeutics Inc has secured US$183m in Series C financing. The investment comes from a consortium of four European investors and Mubadala Capital. Contrary to some reports, more SIRPα modulators exist than the investors initially indicated.
Pharma giant Ipsen will pay US$1bn to acquire French immuno-oncology firm ImCheck Therapeutics SAS. The deal includes US$350m upfront and up to US$650m in milestone payments. ImCheck focuses on butyrophilins, a new class of immune regulators, through its antibody programme ICT01, currently in Phase I trials.


 Adobe stock photos - https://european-biotechnology.com/latest-news/roche-moves-obesity-asset-ct-388-into-phase-iii/
Adobe stock photos - https://european-biotechnology.com/latest-news/roche-moves-obesity-asset-ct-388-into-phase-iii/ NATO
NATO GSK
GSK adobe stock photos - Michael Derrer Fuchs
adobe stock photos - Michael Derrer Fuchs  Adobe stock photo - tippapatt
Adobe stock photo - tippapatt  adobe stock photos - Jiraporn
adobe stock photos - Jiraporn 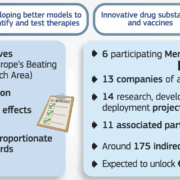 IPCEI Med4Cure
IPCEI Med4Cure adobe stock photos - Miha Creative
adobe stock photos - Miha Creative  Pleiotrope - https://commons.wikimedia.org/w/index.php?curid=15999847
Pleiotrope - https://commons.wikimedia.org/w/index.php?curid=15999847 Ipsen SA
Ipsen SA