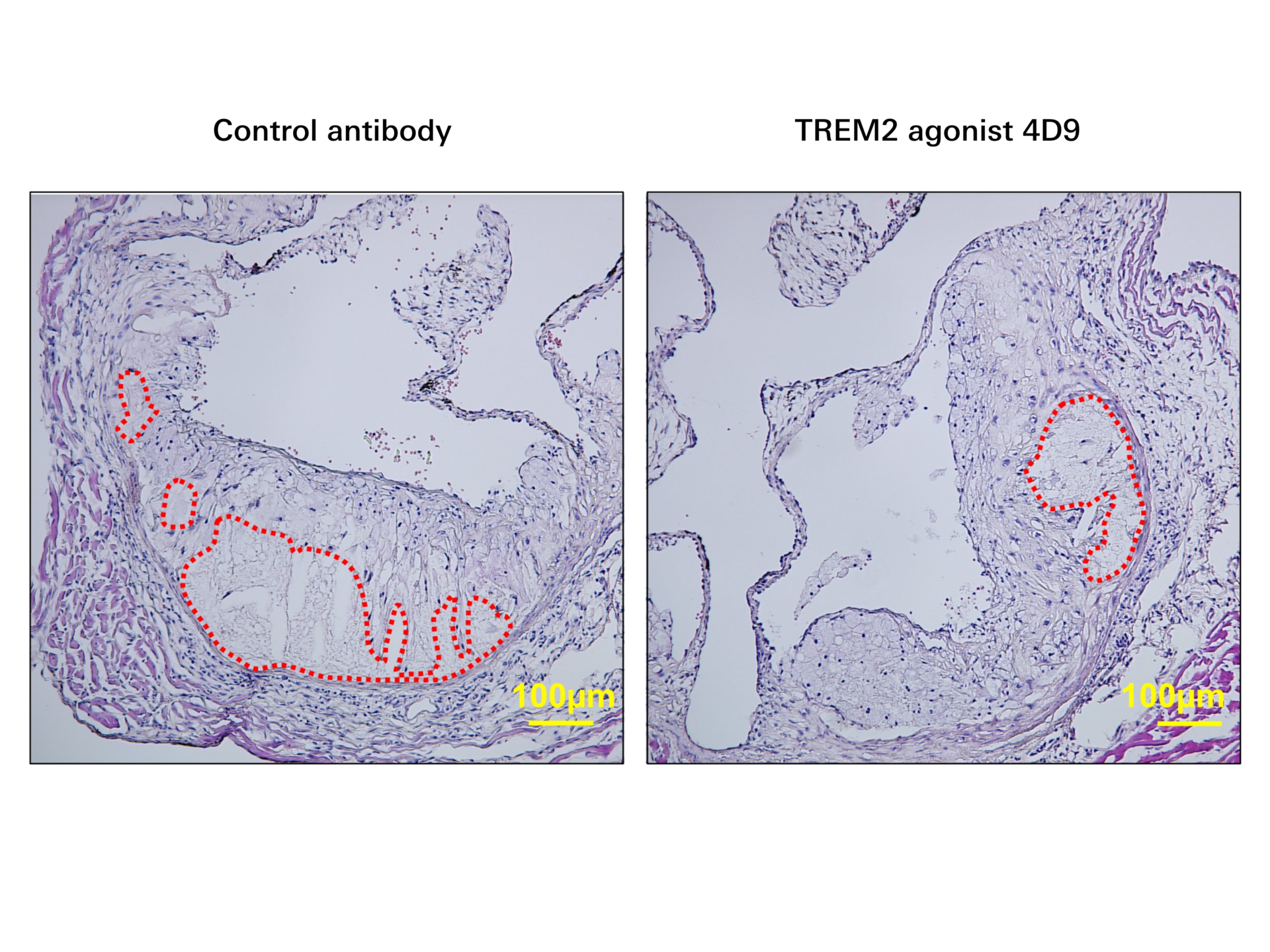
Researcher find potential target for atherosclerosis
Researchers have found a new way to reverse atherosclerotic plaque formation at the early stages. In Nature Cardiovascular Research, Alma...

Swiss vaccine developer Limma Tech get CARB-X grant
LimmaTech said, it will use the grant to advance preclinical development of its hexavalent vaccine candidate targeting Neisseria gonorrheae. Using...
AstraZeneca to take over Fusion Therapeutics Inc
Fusion Pharmaceuticals Inc‘s acquisition for US$2bn upfront marks a major step forward in AstraZeneca delivering on its ambition to transform cancer...

Cambridge university spin-out HutanBio Ltd bags £2.25m seed financing
HutanBio, a Malaysian spin-out from scientists at Cambridge University, UK, has bagged a first tranche of seed investment from London-based Clean...
£26m for Cystic Fibrosis Drug Trial
With funding led by Panakes Partners and involvement from current investors, Enterprise Therapeutics Ltd. has received £26m (~€30.5m) for the Phase...
PulseSight to disrupt AMD space
Aiming to address the pressing challenges of age-related macular diseases (AMD), including wet and dry AMD, along with geographic atrophy (GA),...

Spanish plant-based protein producer Heüra Foods closes Series B financing
Barcelona-based food tech start-up Heüra Foods Srl has cashed in €40m in a Series B financing round supported by Dutch Upfield Holdings BV, Unovis...

