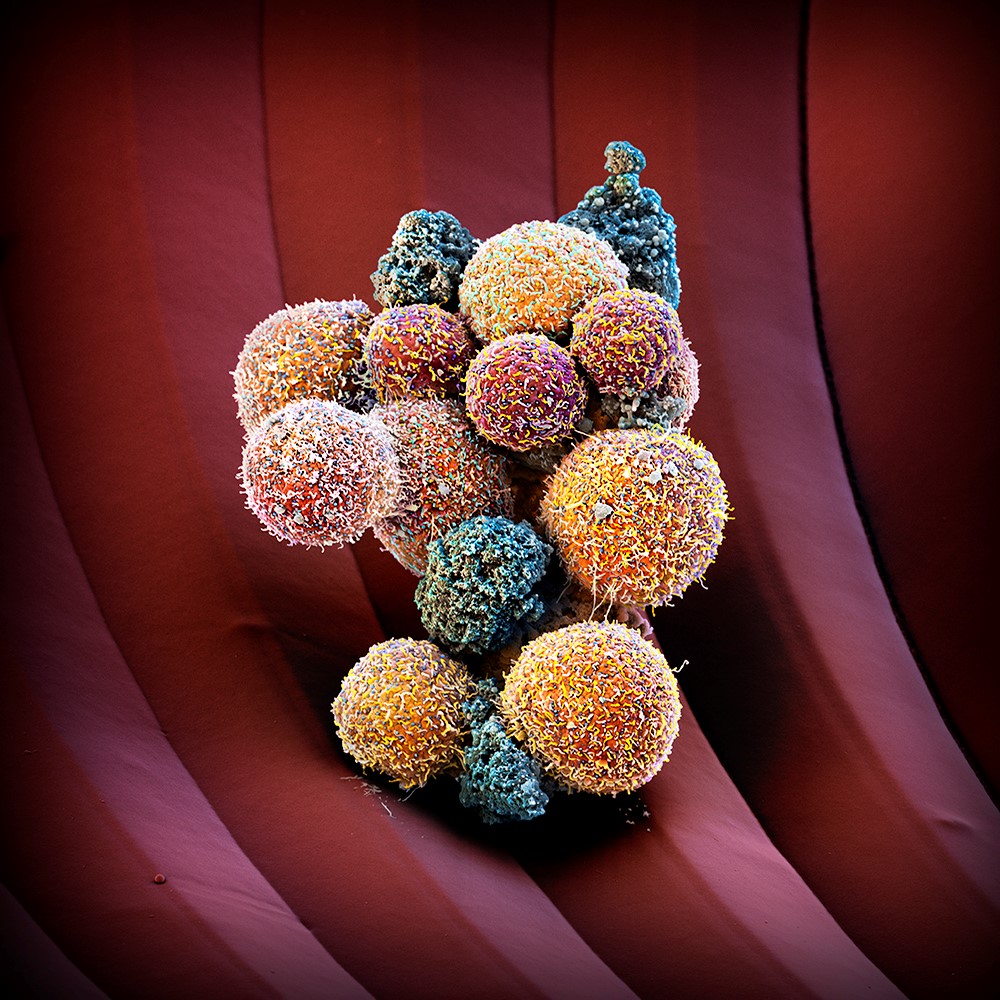
Drug dissociates metastasis precursors in blood
A team of Swiss researchers has identified Na+/K+-ATPase blockers as candidate drugs capable to prevent formation of metastases. In blood, the repurposed drugs identified in a screening dissociated clusters of circulating tumour cells (CTCs), forerunners of metastases.
The development of metastasis is responsible for more than 90% of cancer-related deaths, and patients with metastatic disease are considered incurable. Until recently, it was not possible to isolate and analyse the rare CTCs. However, advances in microfluidic technologies driven forward in projects like Cancer-ID etc are allowing the isolation and characterization of human circulating tumor cells (CTCs) that escaped a primary tumor mass and are in the process of seeding a distant metastasis We thought of acting differently from standard approaches, and sought to identify drugs that do not kill cancer cells, but simply dissociate them, states Nicola Aceto, holder of an ERC starting grant and SNSF professorship. Aceto’s team found CTC cluster formation to be accompanied by hypomethylation of stemness- and proliferation-associated transcription factors that facilitate metastasis seeding. In fact, hypomethylated CTC clusters mimic some properties of embryonic stem cells, including their ability to proliferate while retaining tissue-forming capabilities. The good news, however, from genome-wide DNA methylation analyses of CTC clusters from mice and women with breast cancer was that these epigenetic changes are fully reversible upon the dissociation of the tumours cell clusters in the blood.
According to Aceto, the next goal is now to start clinical trails with the optimised drug candidate. In 2003, the US company Bionaut Pharmaceuticals filed a patent for Na+/K+-ATPase inhibitors to treat pancreatic cancer. The last record of the company on the Internet dating back to 2005 is about a collaboration with the National Cancer Institute to assess if the patented drug can prevent metastases.



 SONUVO
SONUVO Novartis Group
Novartis Group