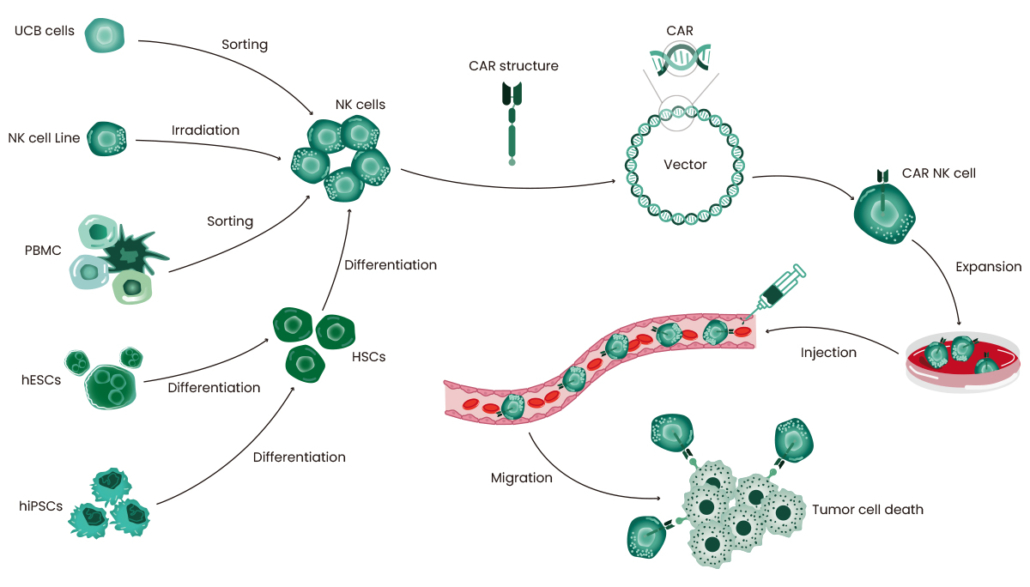
CAR-NK Therapy: An Effective Strategy for Cancer Treatment
CAR-T therapies represent a new successful strategy for the treatment of hematological malignancies; however, recent studies have revealed some limitations, including the occurrence of graft-versus-host disease (GVHD), neurotoxicity, and cytokine release syndromes (CRS). This has prompted researchers to look for alternative, safer cells, such as natural killer (NK) cells 1. NK cells are lymphocytes that kill tumor cells and virus-infected cells non-specifically, without prior sensitization. CAR-NK therapy involves the modification of NK cells with mature chimeric antigen receptor (CAR) technology to exploit their unique target cell recognition mechanism and broad tumor-killing ability. Figure 1 illustrates the CAR-NK therapeutic strategy 1. Compared with CAR-T therapy, CAR-NK therapy has numerous advantages, such as lower GVHD and significantly reduced CRS toxicity and immune effector cell-associated neurotoxicity syndrome toxicity, which enhance overall safety 2, 3. In addition, NK cells have multiple tumor recognition sites, thus potentially reducing antigenic escape failure 4.
CAR Molecule Design
CAR has the ability to specifically recognize target cells and activate NK cells to kill tumor cells. Therefore, the design and engineering of the CAR structure are crucial for CAR-NK therapeutic success. Similar to CAR-T, CAR is composed of an extracellular antigen binding domain, a hinge domain, a transmembrane (TM) domain, and an intracellular domain2. The extracellular domain contains a single-chain variable fragment (scFv) derived from monoclonal antibodies that provides the ability to specifically recognize and bind to tumor antigens. The hinge domain connects extracellular domain to TM domain 5. The intracellular signaling domain determines the strength of the activation signal and directly affects the killing effect 6. Table 1 summarizes the molecules of CAR structure in CAR-NK 2.
Table 1. Molecules of the CAR structure in CAR-NK
| Domains | Molecules |
| Extracellular domain | VH-VL, VL-VH, VH-only |
| Hinge domain | CD8a, CD28, DAP12, IgG4, IgG2 CH2-CH3, IgG1 CH2-CH3, IgG4 CH2-CH3 |
| TM domain | CD3ζ, CD28, CD8a, 4-1BB, DAP12, TCR ab, CD28-CD3ζ, FceRIγ, murine CD3ζ |
| Intracellular domain | CD3ζ, CD28, 4-1BB |
Currently, there are four generations of CARs, with different structures and compositions (Figure 2). First-generation CARs contain only CD3ζ or DAP12 as signaling structural domains. Second-generation CARs express a second signaling structural domain including CD28 or 4-1BB associated with CD3ζ, and third-generation CARs include CD3ζ and two co-stimulatory signaling domains (CD28 and 4-1BB). Finally, fourth-generation CARs involving NK cells can release cytokines, such as IL-2 and IL-15, which regulate NK cells proliferation. This effectively enhanced the anti-tumor activity of NK cells against lymphoma xenografts 1, 7.

Figure 2. The four generations of CAR structure.
(doi:10.1186/s40364-022-00364-6)
Source of NK Cells
NK-92 cell lines have been widely used as a source of CAR-NK cells due to their remarkable proliferation ability in vitro and reduced sensitivity to repeated freeze/thaw cycles. However, as malignant cells from a NK cell lymphoma, NK92 cells have some inherent limitations including potential tumorigenicity risk, absence of CD16 and NKp44 expression, and the need for lethal irradiation prior to infusion, which hampers in vivo expansion potential.
Umbilical cord blood (UCB) presents another attractive source of NK cells with various advantages, such as off-the-shelf availability, low T cell contamination risk, and great potential for proliferation and persistence in vivo. To overcome volume limitations, UCB are expanded to large numbers through a variety of methods covering cytokine mixture and artificial antigen-presenting cells.
In addition, NK cells can be acquired from other sources (as shown in Figure 1), including peripheral blood (PB), cord blood (CB), peripheral blood mononuclear cells (PBMCs), and stem cells such as induced pluripotent stem cells (iPSCs) 8, 9. Scientists are exploring the use of NK cells, and will find the best source for NK-cell immunotherapy for their research.
Preparation and Expansion of CAR-NK Cells
With advances in genetic engineering technology, several methods have been established to generate CAR-NK cells. Commonly used CAR-NK transduction systems utilize viral and non-viral vectors. Viral vectors include retroviruses, lentiviruses and adeno-associated viruses (AAV) 6, with the retroviral transduction being the most common method of delivering CAR to NK cells. Non-viral vectors include the sleeping beauty and PiggyBac transposons. Additionally, mRNA electroporation and the CRISPR/Cas9 technique can also be used.
There are several techniques to expand CAR-NK cells, and the two most commonly used approaches involve feeder cells and cytokine mixtures. Cytokines are important for the in vivo development and survival of NK cells. IL-21 promotes NK cell proliferation and enhances its cytotoxic function. IL-15 stimulates the expansion and cytotoxic activity of NK cells. It is also used in combination with IL-2 to expand NK cells in vitro 10, 11. Sino Biological offers GMP-grade cytokines, including IL-2, IL-12, IL-15, and IL-21, and also provides comprehensive solutions for CAR-NK therapy to support cellular immunotherapy.
Clinical Trials of CAR-NK
As of August 2023, several clinical trials of solid and hematological tumors with CAR-NK therapies have been conducted worldwide. Currently, there are no FDA-approved drugs for CAR-NK and the vast majority are in preclinical phases, with some entering clinical phase I/II (Table 2).
Recently, a CAR-NK therapy was developed to target CD19 for the treatment of B-cell lymphoblastic leukemia/lymphoma in a phase I/II study (NCT05654038). This is a single-center, open-labeled, single-arm, non-randomized investigator-initiated trial to evaluate the efficacy and safety of anti-CD19 universal CAR-NK (UCAR-NK) cells therapy combined with HSCT for the treatment of B-cell hematologic malignancies. In addition, the purpose of another clinical trial (NCT05528341) is to evaluate the safety and effects of NKG2D-CAR-NK92 infusion for the treatment of relapsed/refractory solid tumors.
The clinical trials involving CAR-NK therapy have primarily focused on hematologic malignancies, including B-cell lymphoma, B-cell non-Hodgkin lymphoma (B-cell NHL), acute myeloid leukemia (AML), B-cell hematologic malignancies, acute lymphoblastic leukemia, and multiple myeloma 12-14. CAR-NK therapy is also being tested against solid tumors, such as glioblastoma, breast cancer, small cell lung cancer, colorectal cancer, prostate cancer, and ovarian cancer 11, 15, 16. These clinical trials have shown that CAR-NK therapies target CD19, CD7, CD22, CD33, CD70, BCMA, NKG2D, HER2, DLL3, mesothelin, ROBO1, Claudin 6, or PSMA to achieve therapeutic efficacy 13, 16.
Table 2. Clinical Trials involving CAR-NK cell therapy
| Clinical trial | Target | Disease | Cell source | Status | Phase |
| NCT05182073 | BCMA | Multiple Myeloma Myeloma |
iPSCs | Recruiting | Phase 1 |
| NCT05842707 | CD19 CD70 |
Refractory or Relapsed B-cell Non-Hodgkin Lymphoma |
CB | Recruiting | Phase 1 Phase 2 |
| NCT02944162 | CD33 | Acute Myelogenous Leukemia Acute Myeloid Leukemia Acute Myeloid Leukemia With Maturation |
NK-92 cell line | Unknown | Phase 1 Phase 2 |
| NCT03940833 | BCMA | Multiple Myeloma | NK-92 cell line | Unknown | Phase 1 Phase 2 |
| NCT05654038 | CD19 | B-Cell Lymphoblastic Leukemia /Lymphoma | N/A | Recruiting | Phase 1 Phase 2 |
| NCT04324996 | NKG2D ACE2 |
COVID-19 | CB | Unknown | Phase 1 Phase 2 |
| NCT05645601 | CD19 | Adult Relapsed/Refractory B-cell Hematologic Malignancies |
N/A | Recruiting | Phase 1 |
| NCT05472558 | CD19 | B-cell Non Hodgkin Lymphoma | CB | Recruiting | Phase 1 |
| NCT05667155 | CD19, CD70 | B-cell Non Hodgkin Lymphoma | CB | Recruiting | Phase 1 |
| NCT05008536 | BCMA | Multiple Myeloma, Refractory | CB | Recruiting | Early Phase 1 |
| NCT03692637 | Mesothelin | Epithelial Ovarian Cancer | PBMCs | Unknown | Early Phase 1 |
| NCT05507593 | DLL3 | SCLC, Extensive Stage | N/A | Recruiting | Phase 1 |
| NCT03415100 | NKG2D | Solid Tumor | N/A | Unknown | Phase 1 |
| NCT04847466 | PD-L1 | Gastroesophageal Junction (GEJ) Cancers Advanced HNSCC |
N/A | Recruiting | Phase 2 |
| NCT05528341 | NKG2D | Relapsed/Refractory Solid Tumors | NK-92 cell line | Recruiting | Phase 1 |
| NCT03940820 | ROBO1 | Solid Tumor | Primary NK cells | Unknown | Phase 1 Phase 2 |
| NCT05410717 | Claudin6 | Stage IV ovarian cancer Testis cancer Refractory endometrial cancer |
PB | Recruiting | Phase 1 Phase 2 |
Conclusion
CAR-NK cells have unique advantages over CAR-T cells, such as more precise killing, more cell sources, and greater efficacy against solid tumors. However, some challenges remain, such as cytotoxicity, low transfection efficiency, and storage issues. As CAR-NK therapies continue to be developed and refined, preclinical studies and preliminary clinical data indicate that CAR-NK therapy has improved safety. CAR-NK therapy lacks the human leukocyte antigen matching limitations, possesses a lower GVHD, CRS, and neurotoxicity, in addition to the potential of using allogeneic NK cells as a CAR platform for off-the-shelf therapy. Therefore, CAR-NK therapy holds great promise as a novel cancer treatment strategy with fewer side effects.
References
1. Marofi F, Saleh MM, Rahman HS, et al. CAR-engineered NK cells; a promising therapeutic option for treatment of hematological malignancies. Stem Cell Res Ther. 2021;12(1):374. Published 2021 Jul 2. doi:10.1186/s13287-021-02462-y
2. Pang Z, Wang Z, Li F, Feng C, Mu X. Current Progress of CAR-NK Therapy in Cancer Treatment. Cancers (Basel). 2022; 14(17):4318. Published 2022 Sep 2. doi:10.3390/cancers14174318
3. Valeri A, García-Ortiz A, Castellano E, et al. Overcoming tumor resistance mechanisms in CAR-NK cell therapy. Front Immunol. 2022;13:953849. Published 2022 Aug 3. doi:10.3389/fimmu.2022.953849
4. Liu E, Marin D, Banerjee P, et al. Use of CAR-Transduced Natural Killer Cells in CD19-Positive Lymphoid Tumors. N Engl J Med. 2020;382(6):545-553. doi:10.1056/NEJMoa1910607
5. Fujiwara K, Tsunei A, Kusabuka H, Ogaki E, Tachibana M, Okada N. Hinge and Transmembrane Domains of Chimeric Antigen Receptor Regulate Receptor Expression and Signaling Threshold. Cells. 2020;9(5):1182. Published 2020 May 9. doi:10.3390/cells9051182)
6. Gong Y, Klein Wolterink RGJ, Wang J, Bos GMJ, Germeraad WTV. Chimeric antigen receptor natural killer (CAR-NK) cell design and engineering for cancer therapy. J Hematol Oncol. 2021;14(1):73. Published 2021 May 1. doi:10.1186/s13045-021-01083-5
7. Zhang L, Meng Y, Feng X, Han Z. CAR-NK cells for cancer immunotherapy: from bench to bedside. Biomark Res. 2022;10(1):12. Published 2022 Mar 18. doi:10.1186/s40364-022-00364-6
8. Zhao X, Cai L, Hu Y, Wang H. Cord-Blood Natural Killer Cell-Based Immunotherapy for Cancer. Front Immunol. 2020;11:584099. Published 2020 Oct 22. doi:10.3389/fimmu.2020.584099
9. Xie G, Dong H, Liang Y, Ham JD, Rizwan R, Chen J. CAR-NK cells: A promising cellular immunotherapy for cancer. EBioMedicine. 2020;59:102975. doi:10.1016/j.ebiom.2020.102975
10. Hu W, Wang G, Huang D, Sui M, Xu Y. Cancer Immunotherapy Based on Natural Killer Cells: Current Progress and New Opportunities. Front Immunol. 2019;10:1205. Published 2019 May 31. doi:10.3389/fimmu.2019.01205
11. Kilgour MK, Bastin DJ, Lee SH, Ardolino M, McComb S, Visram A. Advancements in CAR-NK therapy: lessons to be learned from CAR-T therapy. Front Immunol. 2023;14:1166038. Published 2023 May 2. doi:10.3389/fimmu.2023.1166038
12. Huang R, Wen Q, Zhang X. CAR-NK cell therapy for hematological malignancies: recent updates from ASH 2022. J Hematol Oncol. 2023;16(1):35. Published 2023 Apr 7. doi:10.1186/s13045-023-01435-3
13. Shao R, Li Z, Xin H, et al. Biomarkers as targets for CAR-T/NK cell therapy in AML. Biomark Res. 2023;11(1):65. Published 2023 Jun 17. doi:10.1186/s40364-023-00501-9
14. Tian X, Zhang R, Qin H, et al. Immunotherapy of B cell lymphoma with CD22-redirected CAR NK-92 cells. Cent Eur J Immunol. 2023;48(1):1-13. doi:10.5114/ceji.2023.126672
15. Jan CI, Huang SW, Canoll P, et al. Targeting human leukocyte antigen G with chimeric antigen receptors of natural killer cells convert immunosuppression to ablate solid tumors. J Immunother Cancer. 2021;9(10):e003050. doi:10.1136/jitc-2021-003050
16. Khawar MB, Gao G, Rafiq M, et al. Breaking down barriers: The potential of smarter CAR-engineered NK cells against solid tumors [published online ahead of print, 2023 Aug 11]. J Cell Biochem. 2023;10.1002/jcb.30460. doi:10.1002/jcb.30460


 Sino Biological
Sino Biological Sino Biological
Sino Biological