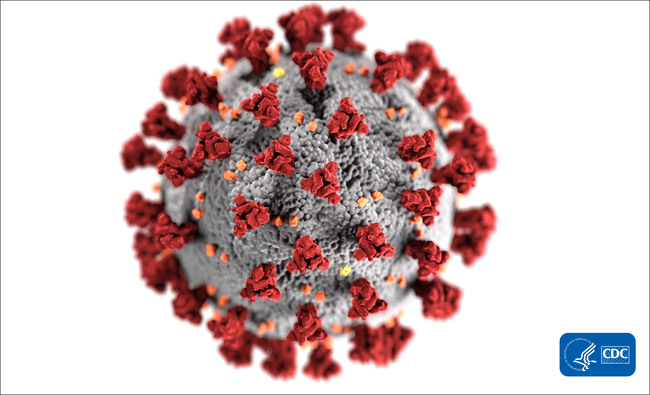
Novacyt inks contract on Covid-19 diagnostics
Paris-based Novacyt SA has signed a global distribution agreement with Bruker Ltd for its COVID-19 diagnostic test.
Novacyt SA ‘s British molecular diagnostics division Primerdesign Ltd (Camberley) signed a global distribution agreement for its COVID-19 test with Bruker-Hain Diagnostics, the molecular diagnostics products business unit of Bruker Corporation. The PCR assay runs on Bruker-Hain Diagnostics’ GenoXtract automated nucleic acid extraction device and is being validated for Bruker’s new FluoroCyclerXT PCR instrument.
Bruker has initiated shipments of the Primerdesign test into Spain, France, Germany and UK. The Company also announces that the Republic of the Philippines Department of Health Food and Drug Administration has approved the Company’s COVID-19 test for commercial use, effective immediately.
Since mid-February,EU labs have increased the capacity of 8,275 diagnostic tests per week to over 7 million tests per week as Roche added a high-throughput test running on its cobas platforms to the bright portfolio of validated Covid-19 PCR tests. However, experts said that diagnostic testing will remain a bottleneck for the next weeks as limitation of COVID-19 spread in EU states willl take some time.



 EyEM, Freepik.com
EyEM, Freepik.com Generare
Generare