MoonLake in US$500m debt deal with Hercules Capital
Swiss MoonLake Immunotherapeutics AG has secured up to US$500m in non-dilutive financing from life sciences debt specialist Hercules Capital to finance the potential 2027 launch of sonelokimab in hidradenitis suppurativa and clinical trials of the IL-17 dimerisation blocker in palmoplantar pustulosis and other inflammatory indications.
MoonLake Immunotherapeutics (Zug, Switzerland) AG’s debt agreement with Hercules Capital Inc. includes an US$75m payment at signature adding to the company’s cash base of US$448m. Further debt tranches will be available upon achievement of pre-specified milestones.
“We were able to secure deal terms, including a very sizeable commitment and a highly competitive cost of capital,” commented Matthias Bodenstedt, Chief Financial Officer at MoonLake Immunotherapeutics. “This financing facility further bolsters our already robust cash position without diluting our shareholders’ stakes and effectively removes any perceived financing overhang as we approach our pivotal data readout for the HS VELA program this summer.”
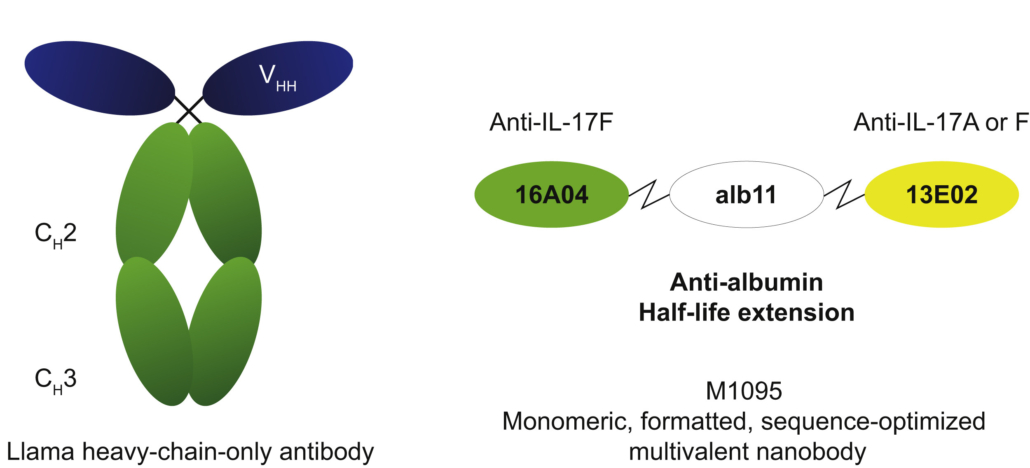
Sonelokimab (M1095) is an investigational ~40 kDa humanised nanobody developed by Ablynx NV and licenced from Merck KGaA in 2021. It consists of three variable regions of heavy-chain-only antibodies domains (VHHs) covalently linked by flexible glycine-serine spacers. With two domains, sonelokimab selectively binds with high affinity to IL-17A and IL-17F, thereby inhibiting the IL-17A/A, IL-17A/F, and IL-17F/F dimers. A third central domain binds to human albumin. According to MoonLake, it facilitates further enrichment of sonelokimab at sites of inflammatory edema.
The company announced it will present interim Phase II results of sonelokimab in palmoplantar pustulosis due to faster enrolment than expected and details of the Phase III testing in hidradenitis suppurativa on 29 April 2025. MoonLake expects the readout of its pivotal study in mid-2025 and expects to launch sonelokimab in 2027 in hidradenitis suppurativa.
Overall, MoonLake is conducting eight late-stage clinical trials in dermatology and rheumatology. Sonelokimab targets so-called AFI diseases in which over-expression of the cytokines IL-17A and IL-17F is a major driver of the pathophysiology. AFIDs include psoriatic arthritis, axial spondyloarthritis (non-radiographic axial and ankylosing spondylitis), hidradenitis suppurativa, psoriasis, palmoplantar pustulosis, generalised pustular psoriasis and pyoderma gangrenosum.


 123rf.com/Paul Grecaud
123rf.com/Paul Grecaud Unsplash+
Unsplash+ Unsplash+
Unsplash+