Vienna is one of Europe’s most dynamic innovation hubs in the life sciences. End of September, the life science community met to celebrate LISAvienna. For 20 years, LISAvienna has been acting as the cluster’s central point of contact. In close collaboration with Austria’s support and funding ecosystem, the platform accompanies and supports bioentrepreneurs through the ups and downs of their journey to transfer innovative business ideas into viable products and services.
ADVERTISEMENT
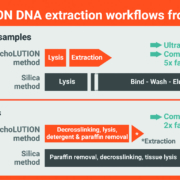
BioEcho Life Sciences develops innovative products that drastically improve the quality, convenience, and speed of standard molecular protocols. The EchoLUTION technology consists of an efficient and gentle lysis, followed by a unique single-step centrifugation, which provides a reduction of procedural steps compared to conventional kits, and considerably reduces plastic waste.
Sino Biological, a biotechnology company listed on the Shenzhen stock exchange subsidiary ChiNext (SZSE: 301047), which provides biological research reagents and related technical contract research services, is pleased to announce the formal signing of a lease with and initiation of construction on its new Center for Bioprocessing (C4B) at its facility in Houston, Texas USA.
Eppendorf Tubes® BioBased reduce the product-specific carbon footprint and support environmental friendly lab workflow.
With its strategic location on the Martinsried campus, the IZB offers an outstanding opportunity to found a company in the vicinity of science and industry.
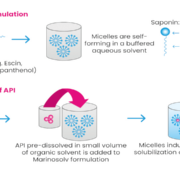
Therapies for autoreactive immune disorders have greatly advanced in the past decades but often have significant side effects. Marinomed Biotech AG strives to improve therapies by increasing the solubility and bioavailability of existing drugs. A proprietary pipeline and Solv4U, a B2B offering for API solubilization, enable the development of clinically meaningful new or improved therapies.
Most people know Switzerland for its watches, mountains, and chocolate. But biopharma professionals know that this country in the heart of Europe offers much more: the ideal setting for innovation and production in the fields of pharma and biotech.
The CRO industry is predicted to grow considerably over the next five years. Trials are becoming more complex, and technology is becoming increasingly interwoven with processes. The CRO of tomorrow must be tech smart, agile, and environmentally conscious to remain relevant. In this changing landscape, how does a CRO cut through the noise in 2022-30?
nearly 200 participants
from 23 countries
30 posters
28 talks
Why are so many vaccine developers choosing Thermo Scientific Charged Aerosol Detection (CAD)? Hear the third very good reasons why CAD was used for analysis of lipid nanoparticles in approved mRNA vaccines.