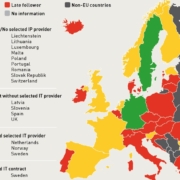
Uncertainty regarding the amount of future funding and political support from the European Commission for the bioeconomy is set to hamper investments into the switch from oil-based industry production to renewable, bio-based manufacture in Europe. The schedule for the review of the bioeconomy strategy has been delayed until the end of 2017. More importantly, it is not yet certain if the strategy will be updated.
ADVERTISEMENT
Drug makers have only until two years from now to make their drugs and packaging counterfeit-resistant. By 9 February, 2019, every prescription drug pack must carry a 2D data matrix code that can be tracked by wholesalers and pharmacists along each stage of the value chain. Additionally, each pack must be sealed with an anti-tampering device. If companies and the NMVOs that handle national databases can’t manage the task, their drugs cannot be sold after the deadline.
Until recently, aroma compounds were either sourced directly from plants or made from petrochemicals with the help of chemical synthesis processes. Now a handful of biotechnological production approaches are making headway and inroads into the key sector. There haven’t been real breakthroughs in big F&F markets yet, but biotech companies look poised to change the industry forever.
Scientists at EPFL in Lausanne have developed a semi-automated technology that may be a game-changer by making the characterisation of the 2,000 DNA-binding proteins much faster, more accurate, and efficient.
To date, insurance business models have always been based on damage control, but that paradigm is changing. Some insurers are now trying to hedge bets by motivating customers to get healthier and track that progress with the help of technology. Critics are concerned about data protection issues, and see this as a first step towards individualised premiums that will erode the principle of solidarity. What are the potential upsides and downsides for customers and societies?