Mitigating regulatory risk with holistic CMC strategies
As advanced therapeutic medicinal products (ATMPs) and other next-generation biologics move toward approval, Chemistry, Manufacturing and Controls (CMC) continues to be the most frequent source of regulatory concern. This article highlights recurring CMC barriers and best practices to mitigate approval delays or refusals.
As more advanced therapeutic medicinal products (ATMPs) and other next-generation biologics seek regulatory approval, the development challenges these products encounter is gaining prominence. Assessments from regulatory authorities consistently show that Chemistry, Manufacturing and Controls (CMC) remains the area of greatest concern.
Last year, the US Food and Drug Administration (FDA) published more than 200 complete response letters (CRLs) that had been issued between 2002 and 2024 for products that have since been approved [1], providing greater transparency into the agency’s decision-making process.
Additionally, FDA has rejected or delayed several new cell and gene therapy (CGT) biologics license application (BLA) submissions due to problems with manufacturing readiness. According to the manufacturers of these products, deficiencies included process control gaps, inadequate stability data, unvalidated analytical methods, and ongoing issues following Good Manufacturing Practice (GMP) inspections [2]
These CMC and GMP issues are well-documented in Europe, where most biological products and all ATMPs go through the Centralised Procedure (CP) with the European Medicines Agency (EMA). The agency has a long-established documentation framework and each product is subject to European public assessment reports (EPARs) [3], whether approved or denied authorization. The release of the CRLs from FDA now allows observers to more rigorously assess areas of respective interest and concern between the health authorities.
Shining a light on CMC
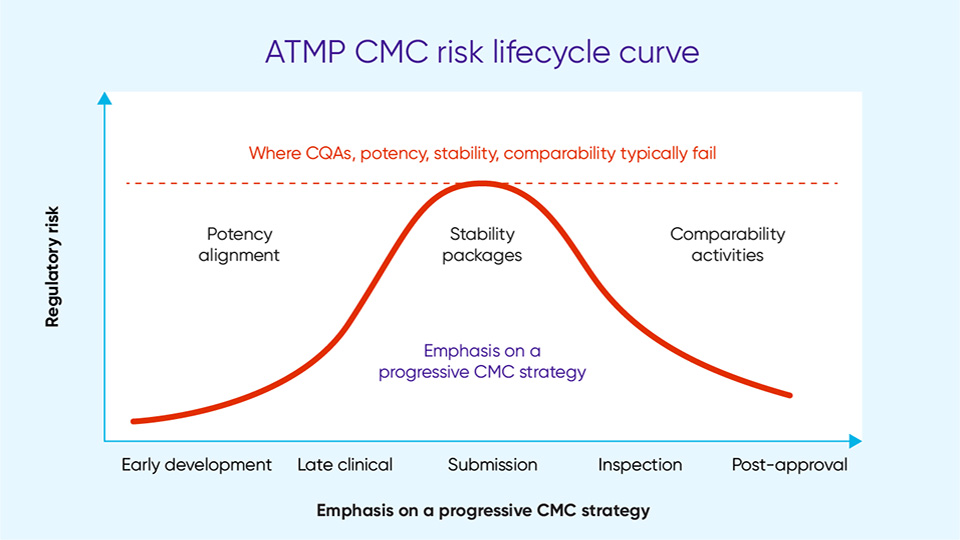
One of the core challenges with developing an ATMP is characterizing the process and getting well-understood critical quality attributes (CQAs.) Having those robustly understood and justified is key to unlocking a lot of the other CMC challenges [4]. A frequently identified CMC issue is potency, particularly getting a relevant potency assay that links to a meaningful CQA that accurately reflects the mode of action of the product. While potency issues are not new, the more complex the therapy, the harder it can be for manufacturers to convince regulators that their CQAs are meaningful and justified and will link back into the clinical efficacy that needs to be demonstrated in clinical trials.
This is relevant not only for potency but also for comparability – another issue highlighted in the CRLs. This refers to the pre- or post-change data when scaling, transferring sites, or changing materials. These changes inevitably occur during product development, particularly with biological products. Unless comparability is linked to meaningful CQAs it is hard to make a compelling case to support those changes.
The same is true for stability. Without stability data linking back to meaningful attributes of the product, it is hard to demonstrate that within reasonable parameters the product is stable over its shelf life and during transfer and transport. Steps to avoid negative findings from the regulators include generating real-time data early, carrying out degradation mapping, and establishing an interim shelf-life supported by a well-justified extension plan.
The CRLs also identify challenges with regards to manufacturers’ control strategies – another issue commonly highlighted in EPARs. They also reinforce the need to have an appropriate manufacturing environment, ensuring that all facilities comply with standard GMP requirements, including sterility assurance. ATMPs are often made in small facilities using novel manufacturing technologies, nonetheless, the same requirements for GMP sterility assurance still apply. Positioning CPP/CQA controls within a risk-based quality framework, such as ICH Q9(R1)5, can demonstrate proactive compliance and inspection readiness.
Manufacturers must ensure they build in all the compendial requirements governed by the European Pharmacopoeia that control sterility and appropriate control and characterization of the process [6].
Taking an inclusive approach
ATMPs are inherently complex products, and small personalized products present even greater challenges. But having the appropriate quality control and processes in place is fundamental to product safety and should be built in from the outset.
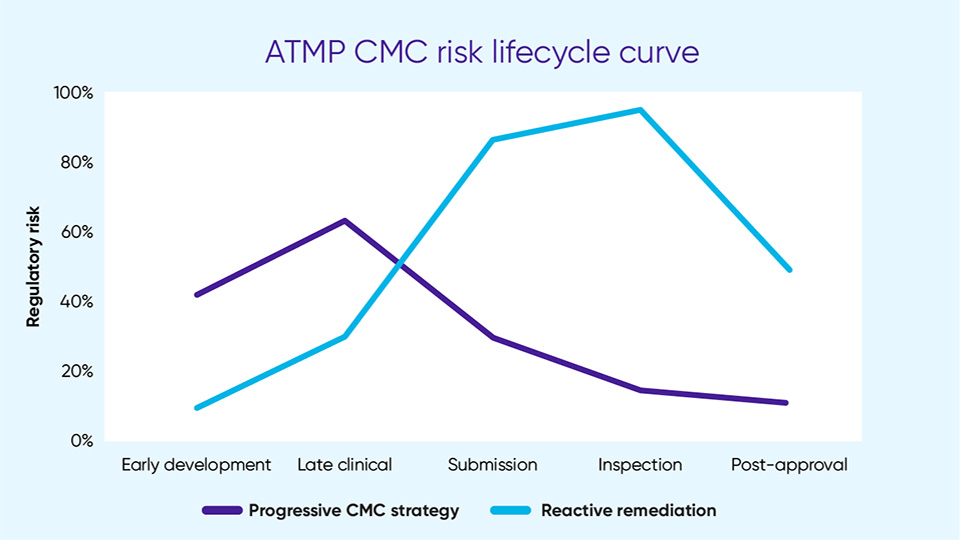
Embedding a holistic approach or best practices into the organization from concept through development and product submission can help to mitigate the CMC issues that have resulted in CRLs in the U.S. and marketing application refusals in Europe. Some best practices manufacturers can put in place include:
- Work cross-functionally to evaluate assays and guide decisions across the full development and commercialization lifecycle.
- Engage the health authorities early to seek scientific advice, leveraging programs like PRIME (where relevant) in Europe, the Innovative Licensing and Access Pathway (ILAP) in the UK, and the Support for Clinical Trials Advancing Rare Disease Therapeutics (START) pilot in the U.S. Raise as many CMC hotspot topics as possible during these interactions.
- Consult key opinion leaders and third-party experts to gain diverse therapeutic-area perspectives and to help build robust plans for monitoring and managing process changes.
- Future-proof the development process to pre-empt evolving regulatory expectations by focusing on data integration (a priority for health authorities) and taking a forward-looking approach to the indicators regulators are looking for before and after inspections.
The CMC challenges facing ATMPs are inherent to the nature of the products and their novelty in the market. As guidelines evolve and as new interpretations of the guidelines emerge, the requirements will become more defined, which will make it easier to identify areas of discrepancy.
By understanding what regulators expect – through assessment of CRLs and EPARs and seeking good scientific advice – manufacturers can better understand this complex and dynamic environment and mitigate risk with their CMC processes.
[1] FDA Embraces Radical Transparency by Publishing Complete Response Letters, FDA, July 2025. https://www.fda.gov/news-events/press-announcements/fda-embraces-radical-transparency-publishing-complete-response-letters
[2] Why gene and cell therapies are stalling at the FDA, Drug Discovery News, Aug 2025. https://www.drugdiscoverynews.com/why-gene-and-cell-therapies-are-stalling-at-the-fda-16527
[3] EMA. European public assessment reports: background and context. https://www.ema.europa.eu/en/medicines/what-we-publish-medicines-when/european-public-assessment-reports-background-context
[4] Johanna I, Daudeij A, Devina F, et al. Basics of advanced therapy medicinal product development in academic pharma and the role of a GMP simulation unit. Immuno-Oncology and Technology, October 2023. https://pmc.ncbi.nlm.nih.gov/articles/PMC10772236/
[5] Q9(R1) Quality Risk Management, FDA, May 2023. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/q9r1-quality-risk-management
[6] European Pharmacopoeia (Ph. Eur.) 11th Edition, European Directorate for the Quality of Medicines & HealthCare. https://www.edqm.eu/en/european-pharmacopoeia-ph.-eur.-11th-edition
Author:
Martin Mewies, Ph.D., is director, regulatory affairs, CMC, at Cencora. He has nearly 30 years of experience in protein biochemistry, with over 20 years in biologics CMC / regulatory.
Disclaimer:
The information provided in this article does not constitute legal advice. Cencora strongly encourages readers to review the references provided with this article and all available information related to the topics mentioned herein and to rely on their own experience and expertise in making decisions related thereto.
This article was originally published in CDMO-Special of European Biotechnology Magazine Spring 2026.