Researchers remyelinate neurons
German neurogenetics at the Max-Planck-Institute for Experimental Medicine have cured the rare incurable hereditary Charcot-Marie-Tooth disease by simply supplementing the food of mice carrying the disease-causing duplication of the PMP22 gene with lecithin. The phospholipid supplementation enabled Schwann cells deficient in lipid synthesis to restore the insulating myelin sheet around peripheral neurons which is needed to speed up neuronal transmission.
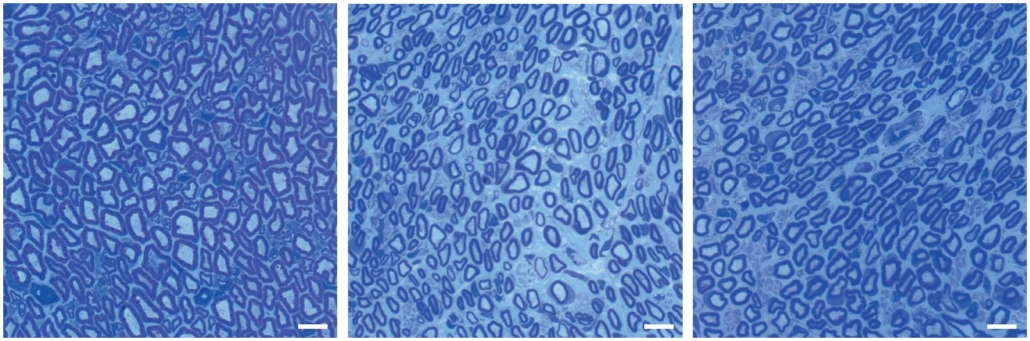
Peripheral nerves of patients with Charcot-Marie-Tooth disease 1A (CMT1A) display aberrant myelination during postnatal development, followed by slowly progressive demyelination and axonal loss during adult life. A research team headed by Michael W. Sereda now demonstrated that myelinating Schwann cells of CMT1A rats carried a transcriptional defect that results in impaired myelin lipid biosynthesis. The distorted stoichiometry of myelin proteins and lipids results in ultrastructural changes of the myelin sheath.
However when the researchers added external phospholipids to the rats feed, the animals overcame the myelination deficit of affected Schwann cells. The complementation of lipids, visualised by flourescence microscopy of labeled phopholipids, rescued the number of myelinated axons in the peripheral nerves of the rats and led to a marked amelioration of neuropathic symptoms. The promising data from the animal testing and especially the already proven compatibility of with humans, promote lecithin as a therapeutic agent for the Charcot-Marie-Tooth disease as well as possibly for similar demyelinating diseases, said Sereda. He now plans to use these findings from animal models in order to help patients by initiating clinical trials.
As the hormone progesterone increases the expression of PMP22 in Schwann cell cultures and in the peripheral nervous system in vivo targeting of the progesterone receptor with antagonists may reduce overexpression of Pmp22 and improve the CMT phenotype without obvious side effects. Thus, the progesterone receptor of myelin-forming Schwann cells is a promising pharmacological target for therapy of CMT-1A that needs further validation in humans.
Charcot-Marie-Tooth (CMT) disease affects more than two million people worldwide. Early symptoms, like walking difficulties or deformed feet can already occur during childhood. Later, muscle muscle weekness in legs and arms occur, but also numbness, prickling or pain. In rare cases, patients may be forced to use wheelchairs. Schwann cells, which surround neurons, normally produce a fatty layer, the myelin, around the axons which allows for a faster transfer of electric signals. In CMT1A diseased Schwann cells show a decreased fat metabolism which causes the impaired myelination.
.


 Planet Volumes via Unsplash+
Planet Volumes via Unsplash+ Valneva
Valneva Servier
Servier