Nanobiotix spins out Curadigm
Cancer-focussed Nanobiotix SA has spun out Curadigm, which offers a platform to improve the bioavailability of nanomedicines by reducing liver and spleen toxicity and accumulation.
"While Nanobiotix remains focused on development of NBTXR3, Curadigm will open new growth pathways for Nanobiotix beyond oncology", said Laurent Levy, CEO of Nanobiotix. "We are thrilled to announce the launch of our subsidiary, Curadigmanother disruptive technology platform we have built as a result of pioneering nanomedicine for more than 15 years."
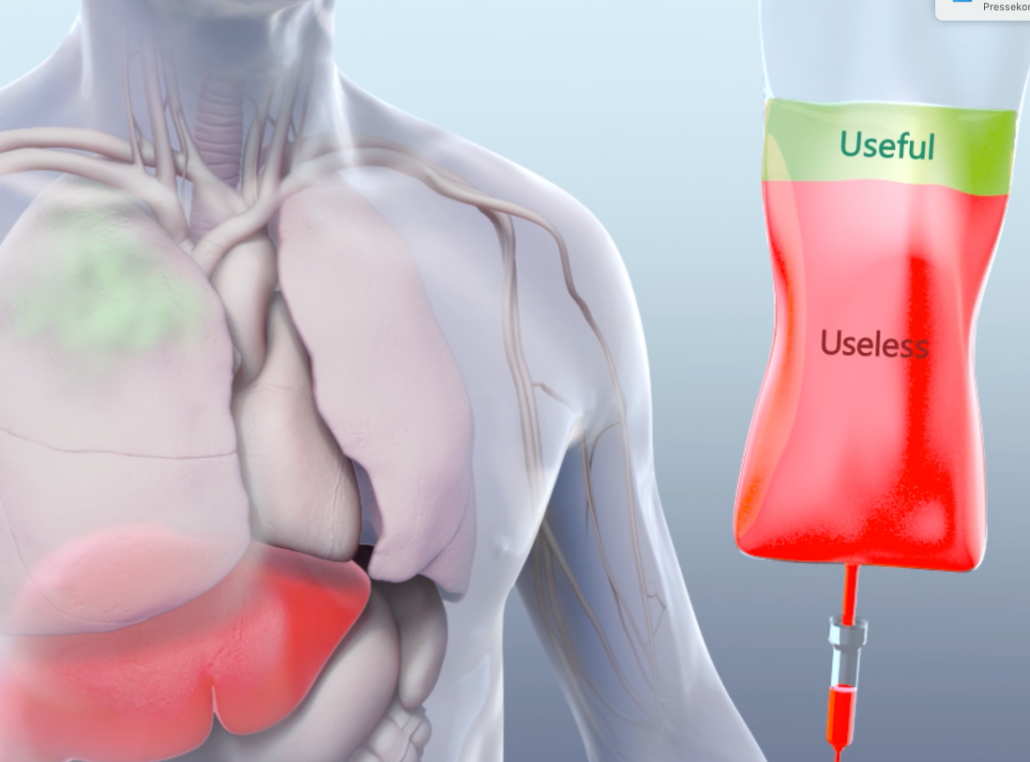
Curadigm’s platform called "Nanoprimers", in fact, could solve a central problem of drug administration: For most nanomedicines, only a small portion of the medicine administered is effective while the rest is cleared from the body without effect or can even be toxic. After injection, the drug moves through the bloodstream, and while a small portion reaches the targeted tissue the remainder is cleared or often accumulates in the liver or spleen.
Curadigm’s Nanoprimer technology has been preclinically shown to improve the bioavailability of medicines by blocking hepatic accumulation or Kupffer cell-mediated drug clearance, i.e. by improving the physico-chemical properties of a drug through liposomal formulation or through application of cytochrome inhibitors that block hepatic clearance systems prior to drug application. Thus, injected prior to the drug candidate, a drug-class specific nanoprimer can increase systemic bioavailability allowing for higher drug concentration in the target tissue, which might affect efficacy at a given dose.
Curadigm is convinced that its technology could have broad implications for healthcare systems due to its potential to increase drug efficacy at a given dose; decrease efficient doses; reduce dose-dependent toxicity and cost, and allow for novel therapeutic approaches
The new company will be led by Dr. Matthieu Germain, who holds deep expertise in the development of nanoparticles after more than 15 years with Nanobiotix, including initial development of the new technology. Dr. Kate Rochlin, leveraging her strong background in early stage company growth, will join the company as Director of Business Development.M. Germain et al., Priming the body to receive the therapeutic agent to redefine treatment benefit/risk profile, SCIENTIFIC REPORTS (2018) 8:4797
- L. Poul et al., Prime the body to receive the treatment: how to improve nanomedicines efficacy, AACR 2019, poster 3613
-Ends-
About NANOBIOTIX: www.nanobiotix.com
Incorporated in 2003, Nanobiotix is a leading, clinical-stage nanomedicine company pioneering new approaches to significantly change patient outcomes by bringing nanophysics to the heart of the cell.
The Nanobiotix philosophy is rooted in designing pioneering, physical-based approaches to bring highly effective and generalized solutions to address unmet medical needs and challenges.
Nanobiotix’s first-in-class, proprietary lead technology, NBTXR3, aims to expand radiotherapy benefits for millions of cancer patients. Nanobiotix’s Immuno-Oncology program has the potential to bring a new dimension to cancer immunotherapies.
Nanobiotix is listed on the regulated market of Euronext in Paris (Euronext: NANO / ISIN: FR0011341205; Bloomberg: NANO: FP). The Company’s headquarters are in Paris, France, with a U.S. affiliate in Cambridge, MA, and European affiliates in Spain and Germany.
About CURADIGM: www.curadigm.com
Curadigm, a wholly-owned subsidiary of Nanobiotix S.A., will be incorporated this year in France and will aim to expand treatment benefits by increasing drug bioavailability while decreasing unintended off-target effects, specifically liver and spleen toxicities.
The Curadigm platform is being developed for use across multiple therapeutic classes to utilize biocompatible nanoparticles to transiently occupy the pathway responsible for therapeutic clearance and hepatic toxicity.
The Curadigm platform is dedicated to bringing broadly applicable delivery technologies to patients to increase efficacy in both current & novel therapeutics and improve patient outcomes.
Contact
https://european-biotechnology.com/up-to-date/latest-news/news/nanobiotix-signs-eur40m-deal-with-eib.html?L=0&cHash=6e95a9aa6183bb7005a03455399da5a0



 CDC/Janice Haney Carr
CDC/Janice Haney Carr 3PBIOVIAN
3PBIOVIAN