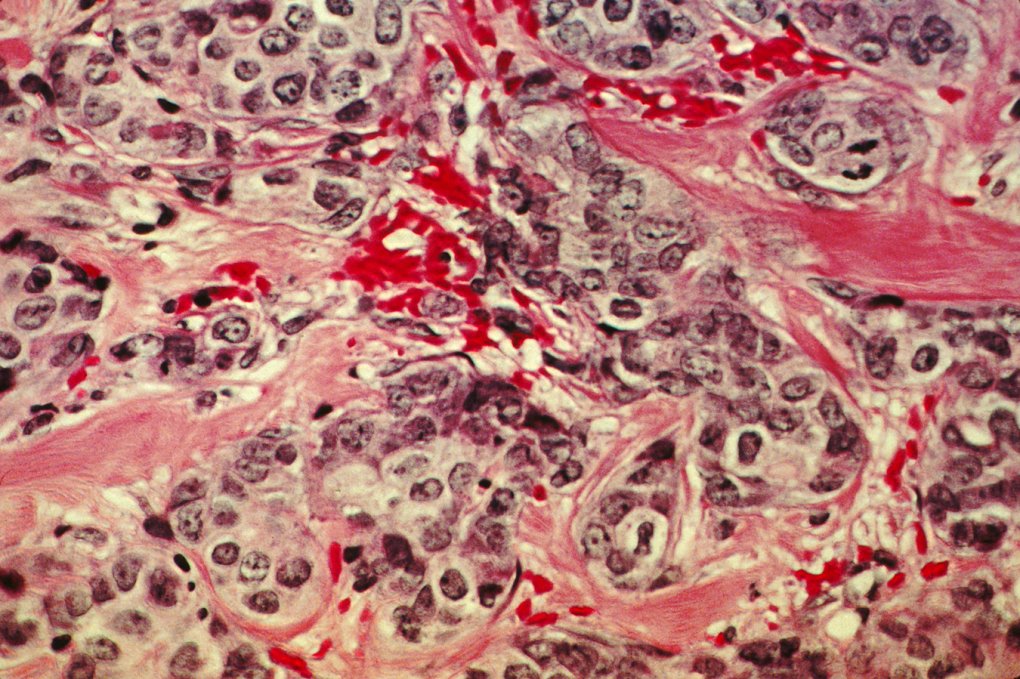
How stress brings cancer to life again
Stress hormones such as cortisol or norephidrine can reawaken dormant tumour cells, a research team led by AstraZeneca has found out.
An international research team led by AstraZeneca’s oncology expert Dmitry I. Gabrilovich report that
that stressed neutrophils might act as a tumuor’s alarm clock in mouse models and lung cancer patients. Inducing stress in mice resulted in tumour cell reactivation, whereas treating mice with ?-blockers to inhibit stress hormone signaling prevented tumour cell reactivation.
Stress hormones and neutrophils may contribute to the recurrence of tumors years after treatment by awakening dormant cancer cells, suggests a study of mice and data from 80 patients with lung cancer. Stress hormones cause rapid release of proinflammatory S100A8/A9 proteins by neutrophils. S100A8/A9 induce activation of myeloperoxidase, resulting in accumulation of oxidized lipids in these cells. Upon release from neutrophils, these lipids up-regulate the fibroblast growth factor pathway in tumor cells, causing tumor cell exit from the dormancy and formation of new tumor lesions. Higher serum concentrations of S100A8/A9 were associated with shorter time to recurrence in patients with lung cancer after complete tumor resection. Targeting of S100A8/A9 or ?2-adrenergic receptors abrogated stress-induced reactivation of dormant tumor cells.
The researchers suggest that beta blockers or compounds that target S100A8/A9 proteins should be evaluated as potential therapies to disrupt the reactivation process, but stress the need for more sophisticated models of tumour cell dormancy.


 Santhera
Santhera