Macrocycles: big is the new beautiful
In the evolving landscape of drug discovery and new design, scientists and pharmaceutical innovators continually strive to develop therapies that are both highly selective and clinically effective, while addressing targets previously deemed “undruggable”. In recent years, macrocycles – a class of large, ring-shaped molecules – have emerged as a compelling solution at the crossroads between traditional small molecules and large biologics, offering a blend of high specificity, rich chemical diversity and promising pharmacological profiles.
What if synthetic large molecules could be designed to fit almost any receptor pocket imaginable? While this may sound like a new frontier in drug development, the underlying idea is far from new. A wide range of protein scaffolds has already been explored and advanced from preclinical research toward the clinic, including Pieris’ lipocalin scaffold in Munich and the DARPin-based molecules developed by Molecular Partners in Zurich. Although Pieris ultimately failed to deliver on the promise of an antibody-like structure with greater flexibility and smaller size, Molecular Partners is now entering the clinic, where the first data will reveal whether this approach can carve out a place in the biologics space. But a new player has now entered the scene, and this time size seems to matter less than ever. Fueled by advances in chemistry, machine learning, and automated discovery platforms, macrocycle drug discovery has surged into the spotlight, attracting major biotech investors, global pharmaceutical companies, and ambitious European startups alike.
What are macrocycles?
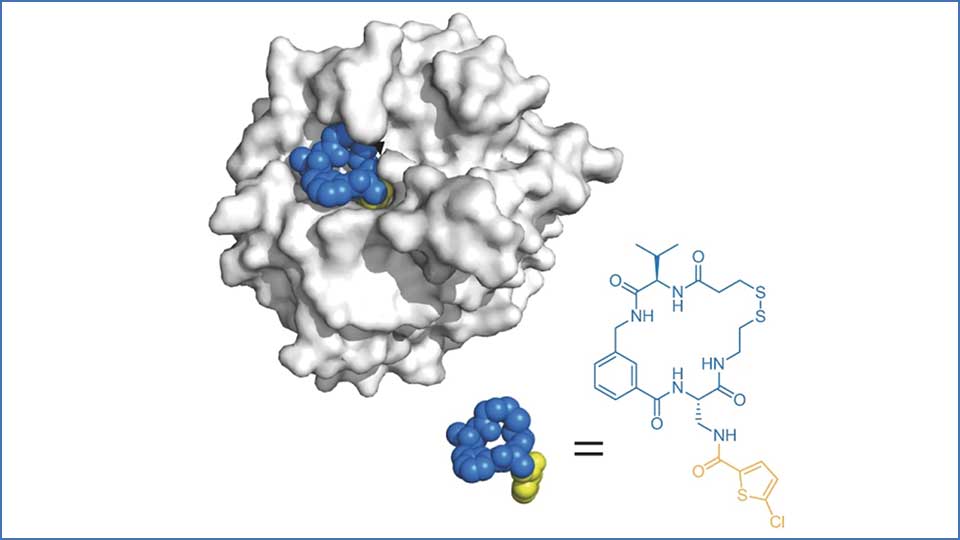
At their core, macrocycles are organic compounds in which the atoms form a large ring structure. Formally, they contain a circle of at least 12 non-hydrogen atoms in a single contiguous loop. This cyclic topology grants them distinct conformational properties compared with simpler linear molecules: they are pre-organised and semirigid, which can dramatically enhance how they bind to biological targets.
Macrocycles cover a diverse chemical space that overlaps neither with traditional “Rule of Five” small molecules (designed for ease of oral absorption) nor with large biologics such as monoclonal antibodies. This hybrid space lets macrocycles combine the high target affinity and selectivity often associated with biologics with the cell permeability and, increasingly, oral bioavailability more typical of small molecules.
This feature makes macrocycles particularly valuable for tackling protein-protein interactions, flat or groove-like binding surfaces, and other challenging targets that have frustrated small-molecule efforts for decades.
Early success and history
Macrocycles are not entirely new to medicine, their therapeutic potential has been recognised for decades. Indeed, some of the earliest and most successful macrocycle drugs were discovered in nature. The immune-suppressant cyclosporine, isolated from a fungus in the early 1970s, transformed transplant medicine by suppressing T-cell activation and helping prevent organ rejection.
Natural macrocyclic compounds and their derivatives have since made their way into numerous approved medicines, particularly in infectious disease and oncology. Yet, historically, most macrocycles approved to date have been natural products or closely related derivatives rather than synthetic designs.
This presents an important nuance: although macrocycles have long been known to be effective drug molecules, designing and synthesising them de novo – especially with properties amenable to oral dosing – has proven difficult. The complexity of macrocycle chemical space made systematic exploration extremely challenging until recently.
Why macrocycles matter today
In the past decade, technological and scientific advances have begun to unlock macrocycles as a practical and versatile drug class. Several factors are converging to propel macrocycle innovation:
Enhanced binding capabilities: Macrocycles can often achieve higher affinity and selectivity for difficult targets than linear small molecules, thanks to their cyclic, pre-organised structures.
Broader chemical space: Modern synthetic and computational methods now allow exploration of vast libraries of macrocycle candidates, making it more feasible to identify compounds with drug-like properties.
Improved oral bioavailability: Many newer macrocycles are being deliberately designed to overcome traditional absorption barriers, bringing oral macrocycle therapies within reach.
Technology platforms and AI: High-throughput synthesis, DNA-encoded libraries and machine learning are now being used to design, generate and screen macrocycle libraries at unprecedented scale, accelerating discovery and reducing risk.
European Frontrunners
Collectively, these innovations are positioning macrocycles not as a niche modality but as a strategic drug class capable of addressing unmet therapeutic needs across oncology, immunology, cardiovascular disease and beyond.
In Europe the two most prominent pioneers in this space are Orbis Medicines, a Copenhagen-based biotech founded in 2021 with backing from Novo Holdings and Forbion, and Curve Therapeutics, founded in Southampton (UK) in 2019.
Orbis has developed a platform called nGen that integrates high-throughput chemistry, large-scale assaying and machine learning to explore macrocycle chemical space systematically. From initial libraries of billions of potential compounds, the platform rapidly synthesises and evaluates hundreds of thousands of candidates to pinpoint those with desirable therapeutic attributes, particularly oral bioavailability and membrane permeability.
The goal at Orbis is to deliver “nCycles” – orally dosable macrocycle drugs – against targets traditionally validated by blockbuster biologics. By targeting both intra- and extracellular proteins, Orbis hopes to broaden the potential disease indications beyond what most small molecules or biologics can achieve. This approach addresses the longstanding challenges of macrocycle design, and, as research published in top journals has suggested, represents a potential leap forward in translating macrocycle science into real medicines.
Curve Therapeutics is harnessing the power of gene-encoded screening, to identify a new generation of therapeutics to address a broad range of intracellular targets. Curve’s macrocyclic platform involves screening Microcycle® libraries (vast collections of small cyclic peptides) inside mammalian cells, resulting in the discovery of functional hits against targets in their dynamic and pharmacologically relevant forms. Microcycles are compact, rigid macrocycles which behave like small molecules, allowing for the discovery of novel, and more effective drugs.
Curve completed its £40.5 million Series A financing in 2024, and has since made significant progress in advancing its proprietary drug discovery platform, and establishing a pipeline of potential first-in-class programmes. Curve had also received earlier validation on the potential of its Microcycle platform through a collaboration with MSD worth up to $1.7 billion, cementing its status as an established pioneer in the field.
The transition from an academic start up to a recognised and visible player in the field has clearly accelerated with the investment from well-established institutions. The UK biotech secured £40.5 million in Series A funding from investors including Advent Life Sciences, Epidarex Capital, Pfizer Ventures, Columbus Venture Partners and British Business Bank. The capital has enabled the company to strengthen its leadership team with several senior appointments, including Simon Jones as CFO/COO, Andre Hoekema as chair of the board, Cora Griffin as head of business development and Rab Prinjha as chief R&D officer.
Curve is building its strategy around its proprietary Microcycle platform, developed from two decades of research led by co-founder and CSO Professor Ali Tavassoli. The technology enables high-throughput screening of drug candidates directly in living mammalian cells, allowing for the evaluation of biological activity in a more physiologically relevant setting than conventional discovery methods. The company believes this approach could help address one of the persistent challenges in drug discovery: targeting intracellular proteins that remain difficult to access with existing modalities such as antibodies.
Curve’s internal pipeline initially focuses on intracellular oncology and I&I targets, with proceeds from the Series A round earmarked to advance these programmes towards clinical development. Looking ahead, the company aims to expand both its internal pipeline and partnered programmes while securing additional pharmaceutical collaborations.
Global macrocycle momentum
Momentum in the macrocycle field is not limited to Europe. Major pharmaceutical companies are actively partnering with innovative macrocycle biotechs to build next-generation pipelines.
One of the most high-profile examples is the partnership between Swiss pharma giant Novartis AG and California-based macrocycle specialist Unnatural Products (2026). Under this agreement, Novartis has licensed macrocyclic peptide programmes aimed at cardiovascular disease, paying upfront and milestone fees that could total up to US$1.7 billion, with tiered royalties on future sales.
Unnatural Products – which integrates AI and advanced chemistry to generate macrocyclic peptides with enhanced permeability and drug-like profiles – has also secured partnerships with other global players. In 2025, for example, it agreed a sizeable collaboration with argenx (Amsterdam) to develop drugs for challenging immunology targets.
These collaborations reflect a broader industrial recognition that macrocycles are an emerging modality with strategic value, capable of complementing and, in some cases, extending beyond traditional biologics and small molecules.
Together, these features make macrocycles well suited for difficult targets, broad therapeutic areas and complex disease biology, which are areas where existing modalities offer limited potency, specificity or patient convenience.
Opportunities and challenges
The combination of platform technologies, AI-driven design, and strategic industry partnerships suggests that macrocycles may soon become a mainstream therapeutic modality.
With oral macrocycles now appearing in late-stage clinical programmes and major pharma backing collaborations, the next decade could see these molecules realise their long-heralded promise in a wide range of diseases.
This article was originally published in European Biotechnology Magazine Spring 2026.


 Bayer
Bayer
 Getty Images, for Unsplash+
Getty Images, for Unsplash+