
Pierre Fabre SAS in US$31m licence deal with Kinnate Biopharma Inc
With the US$31m agreement Pierre Fabre licences the global rights to exarafenib and other pan-RAF program assets from Kinnate...
Seabelife SAS to develop next-gen dry AMD drug
French drug developer SeaBeLife SAS has bagged a €1.5m fund within the i-Nov 2024 innovation competition, which is part of the French government’s...
Senisca Ltd raises £3.7m seed money
The proceeds of Senisca’s latest funding round led by Lifespan Vision Venture will support the preclinical development of the company’s...

Pacifico Biolabs raises US$3.3m in pre-seed round
Riding the wave of mushroom mycelium-based foods, which are advertised as a climate- and animal-friendly alternative to meat, Pacifico Biolabs GmbH...

Novozymes and Chr. Hansen Complete Merger, Forming Novonesis
In a significant development in the biosolutions sector, Novozymes A/S and Chr. Hansen Holding A/S successfully completed their merger, forming a new...

New law to speed up clinical trials
The draft Medical Research Act (Medizinforschungsgesetz) presented by Health Minister Karl Lauterbach in Berlin before Easter is the centrepiece of...
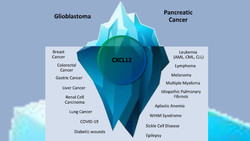
TME Pharma gets FDA Fast Track for Glioblastoma
TME Pharma N.V. with research sites in France and Germany (listed at Euronext Growth Paris: ALTME) is developing novel therapies for treatment of...

