
Amsilk partners with 21st.BIO
Advanced materials specialist AMSilk GmbH (Neuried, Germany) and biotech production specialst 21st.BIO (Copenhagen) have announced to join forces to...
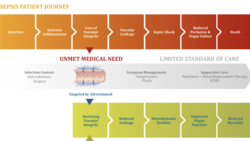
Shock: Adrenomed AG gets FDA fast track designation
After Adrenomed AG has demonstrated that its septic shock treatment led to a 60% reduction in relative 28-day mortality vs. placebo in a stratified...
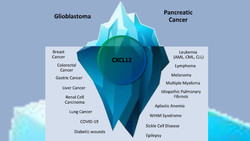
TME Pharma gets FDA Fast Track for Glioblastoma
TME Pharma N.V. with research sites in France and Germany (listed at Euronext Growth Paris: ALTME) is developing novel therapies for treatment of...

Bayer's Ask Bio unit starts Phase II heart failure gene therapy
The Phase II trial called GenePHIT is the largest adaptive, double-blind, placebo-controlled, randomised, multicentre trial ever to evaluate the...

Roche opens €90m gene therapy development centre
Roche sees the €90m investment as a harbinger of the German government's "National Strategy for Gene and Cell Therapies", which has been...

Roche jumps ship on AC Immune
In recent trials, both Semorinemab, a monoclonal antibody (mAb) binding all forms of Tau, and Crenezumab, a mAb targeting multiple forms of misfolded...

Novozymes and Chr. Hansen Complete Merger, Forming Novonesis
In a significant development in the biosolutions sector, Novozymes A/S and Chr. Hansen Holding A/S successfully completed their merger, forming a new...

