
Pierre Fabre SAS in US$31m licence deal with Kinnate Biopharma Inc
With the US$31m agreement Pierre Fabre licences the global rights to exarafenib and other pan-RAF program assets from Kinnate...

Bayer SE secures rights to in €310m deal
Under the licence deal, BridgeBio Pharma, Inc. will pass the commercialisation rights for the transthyretin stabiliser acoramidis in Europe where...
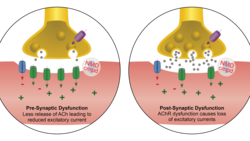
NDM Pharma provides POC for NMD670
In Science Translational Medicine the team headed by Thomas Holm Pedersen from NDM Pharma A/S report that they have conducted animal studies and a...

Ipsen puts US$1.8bn in pipeline expansion in movement disorders
Under the agreement, Skyhawk will provide its discovery platform for RNA-targeting small molecules across several therapeutic areas, including rare...

Cambridge university spin-out HutanBio Ltd bags £2.25m seed financing
HutanBio, a Malaysian spin-out from scientists at Cambridge University, UK, has bagged a first tranche of seed investment from London-based Clean...

Curve Therapeutics raises £40.5m in Series A financing
New investors Columbus Venture Partners and British Patient Capital and founding investor Advent Life Sciences as well as seed investor Epidarex...
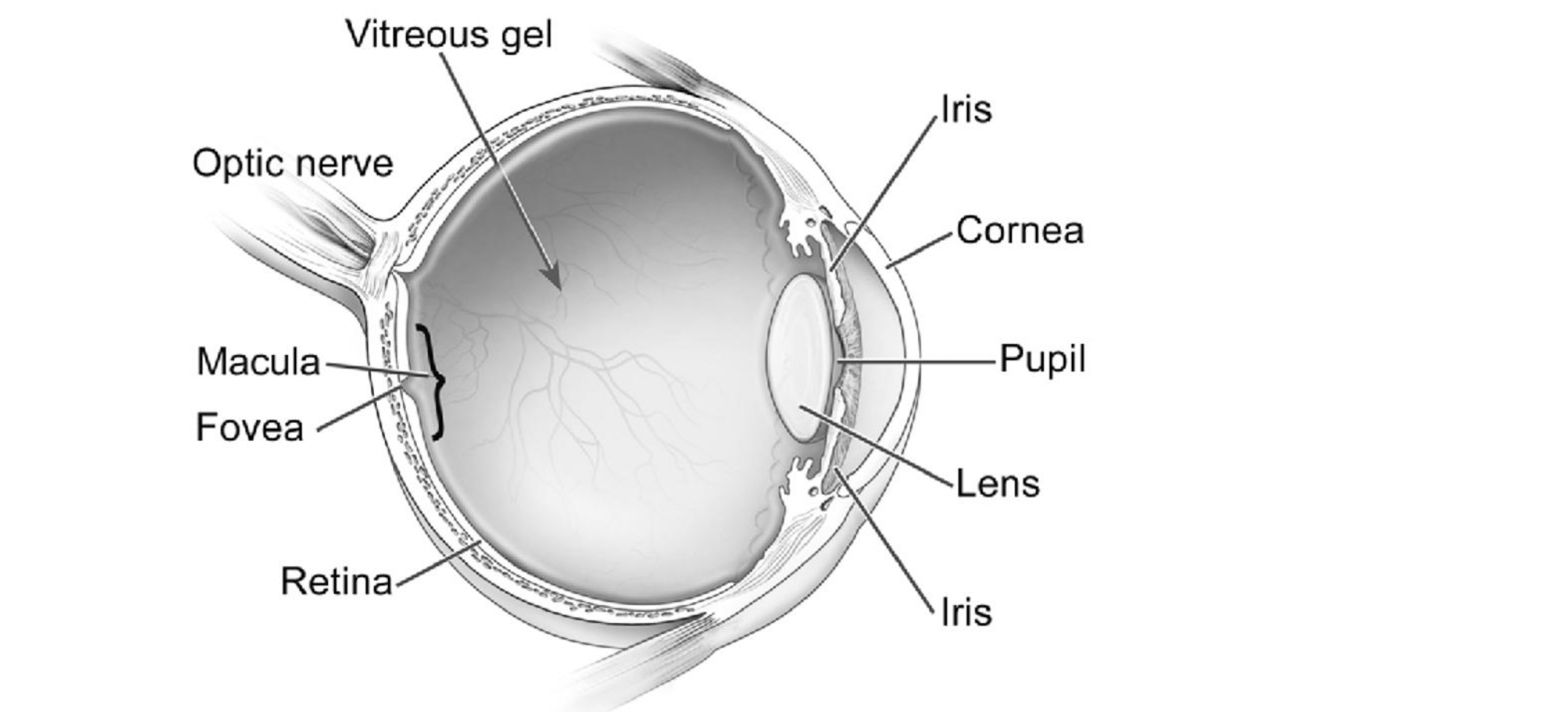
Seabelife SAS to develop next-gen dry AMD drug
French drug developer SeaBeLife SAS has bagged a €1.5m fund within the i-Nov 2024 innovation competition, which is part of the French government’s...

