
AMPLY Discovery Ltd bags £1.4m funding to tackle complex diseases
The 2021 spin-out from the Queen’s University Belfast announced it will close a seed financing round this year.vAMPLY is speeding up the discovery of...

Danish 21st.BIO A/S opens multipurpose production plant
Today, EU Executive Vice-President Margrete Vestager launched a multi-purpose pilot production facility in Copenhagen, with which 21st.BIO A/S aims...
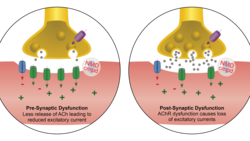
NDM Pharma provides POC for NMD670
In Science Translational Medicine the team headed by Thomas Holm Pedersen from NDM Pharma A/S report that they have conducted animal studies and a...

German companies to revolutionise lab plastics market
Plastic consumables used in pharmaceutical and biotechnological research must meet high quality standards. A German start-up has now launched the...
Lonza and Oxford Nanopore collaborate to advance mRNA Therapeutics
The significance of mRNA technology has been highlighted in recent years, particularly with its role in rapidly developing and bringing COVID-19...

Novozymes and Chr. Hansen Complete Merger, Forming Novonesis
In a significant development in the biosolutions sector, Novozymes A/S and Chr. Hansen Holding A/S successfully completed their merger, forming a new...

Bayer SE secures rights to in €310m deal
Under the licence deal, BridgeBio Pharma, Inc. will pass the commercialisation rights for the transthyretin stabiliser acoramidis in Europe where...

