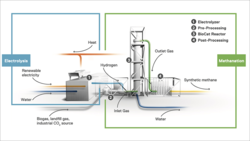
Electrochaea sells biomethane reactor to Again A/S
Like German bio-CH4 producer Electrochea GmbH, Danish Again A/S is using CO2-fxing archaea, but to manufacture carbon-negative chemicals such...
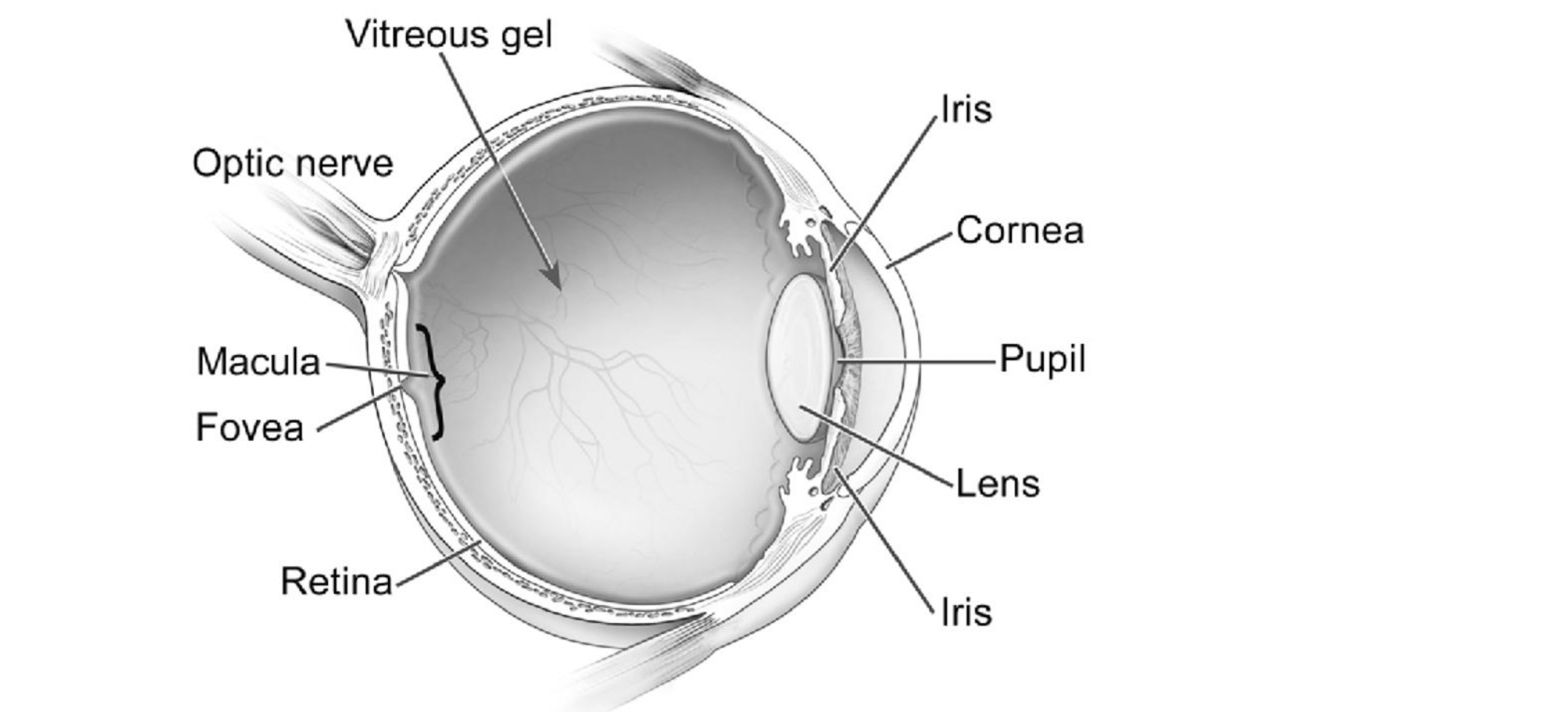
Seabelife SAS to develop next-gen dry AMD drug
French drug developer SeaBeLife SAS has bagged a €1.5m fund within the i-Nov 2024 innovation competition, which is part of the French government’s...
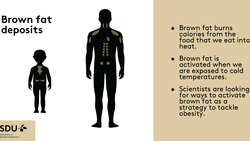
Obesity: reseachers switch on brown fat cells
The new discovery could herald a revolution in obesity research. If the principle discovered in male mice also applies in humans, therapeutics...
Innocent Meat bags €3m financing
The three-year-old startup announced it will use the new proceeds to optimise components of its automated plug-and-produce solution for mass...
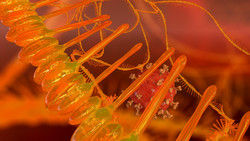
Lonza and Oxford Nanopore collaborate to advance mRNA Therapeutics
The significance of mRNA technology has been highlighted in recent years, particularly with its role in rapidly developing and bringing COVID-19...

Curve Therapeutics raises £40.5m in Series A financing
New investors Columbus Venture Partners and British Patient Capital and founding investor Advent Life Sciences as well as seed investor Epidarex...

Solar Foods starts Factory01
Following market authorisation in Singapore two years ago, the Finnish company Solar Foods Oy and the Japanese food giant Ajinomoto Group want to...

