Swiss Santhera Pharmaceuticals EU-approved LHON treatment Raxone (idebenone) has failed to show clinical efficacy in another indication: After US and EU regulators rejected an extension of market approval in Duchenne muscular dystrophy last September, an NIH-sponsored trail now showed no difference to placebo treatment in patients with primary progressive multiple sclerosis (PPMS).
It seems not to be a good message for company’s the share value, which had went down from CHF70.95 in September to CHF25.85 this Friday. The exploratory Phase I/II clinical trial in 77 PPMS patients, which were randomised 2:1, showed no difference between patients, who received a daily 2,25mg dose and those receiving placebo. However, the safety profile to idebenone was confirmed.
The trial combined a one-year observational pre-treatment phase, followed by a two-year placebo-controlled intervention period. There was no difference in the occurrence and severity of adverse events between the treatment groups indicating that idebenone at a daily dose of 2,250 mg was well tolerated.
The primary outcome to explore the efficacy of idebenone was the change in the CombiWISE, a new rating scale. Top-line analysis of the CombiWISE data and other clinical assessments and biomarkers (such as the disability progression scale EDSS-plus and changes in ventricular volume) indicate that there was no difference between treatment groups for measures of disease progression.
"The long-term study in patients with PPMS confirms the favorable safety profile of idebenone given at higher dose than the currently approved dose for Raxone® in Leber's hereditary optic neuropathy," said Thomas Meier, PhD, CEO of Santhera. "We thank the NIH team for conducting this long-term pilot study which will add to the knowledge of disease progression and data collection instruments. Clearly, the small sample size is a limitation when studying a therapeutic intervention in such a complex, relentlessly progressing neurological disease."
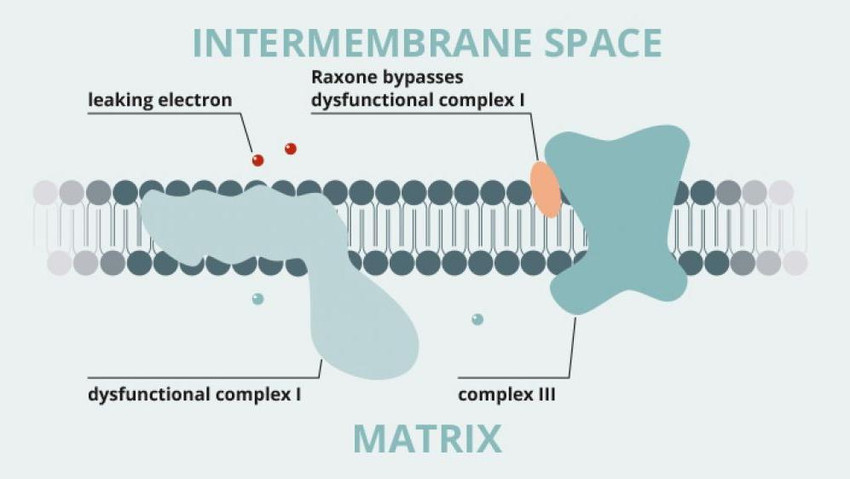
Santhera's Raxone® (idebenone) was authorised under exceptional circumstances in 2015 in the European Union, Norway, Iceland, Liechtenstein and Israel for the treatment of visual impairment of patients with the rare eye disease Leber's hereditary optic neuropathy (LHON). Last September, the European Medicine Agency’s CHMP rejected a type II extension application to the company’s coenzyme Q10 analogon as treatment for patients with Duchenne muscular dystrophy (DMD). Previously, the US FDA rejected market authorisation in this indication requesting further clinical data. The company’s shares dropped by 58% to CHF38.50. At the end of January 2018, the CHMP finally rejected an appeal its negative opinion for use in treating Duchenne muscular dystrophy (DMD). As Santhera’s most advanced product candidate has first to complete a 3-year clinical study in the US to potentially receive FDA approval in this indication, commercialisation of the drug in lagging behind the original plans of the drug maker.
Raxone is a ‘hybrid medicine’. This means that it is similar to a ‘reference medicine’ containing the same active substance, but Raxone contains idebenone at a different strength. The reference medicine for Raxone is Mnesis.