
Swiss vaccine developer Limma Tech get CARB-X grant
LimmaTech said, it will use the grant to advance preclinical development of its hexavalent vaccine candidate targeting Neisseria gonorrheae. Using...
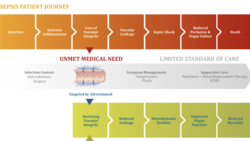
Shock: Adrenomed AG gets FDA fast track designation
After Adrenomed AG has demonstrated that its septic shock treatment led to a 60% reduction in relative 28-day mortality vs. placebo in a stratified...

Bayer's Ask Bio unit starts Phase II heart failure gene therapy
The Phase II trial called GenePHIT is the largest adaptive, double-blind, placebo-controlled, randomised, multicentre trial ever to evaluate the...

EU parliament backs deregulation of New Genome Techniques
With the MEPs' approval of the amendments to the European Commission's draft legislation, the way is now clear for a regulation that distinguishes...
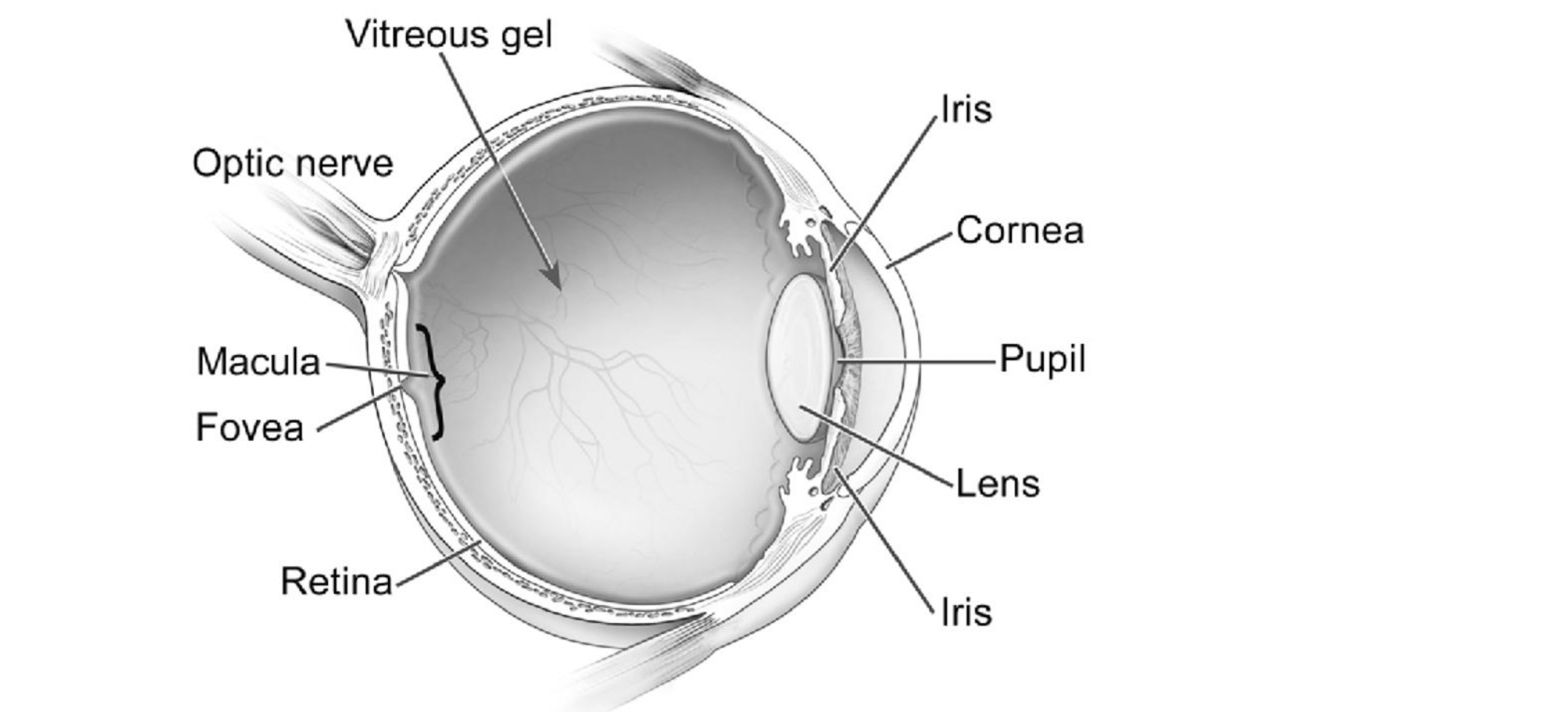
SeaBeLifeSeabelife SAS to develop next-gen dry AMD drug
French drug developer SeaBeLife SAS has bagged a €1.5m fund within the i-Nov 2024 innovation competition, which is part of the French government’s...
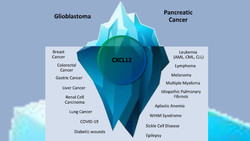
TME Pharma gets FDA Fast Track for Glioblastoma
TME Pharma N.V. with research sites in France and Germany (listed at Euronext Growth Paris: ALTME) is developing novel therapies for treatment of...

Amsilk partners with 21st.BIO
Advanced materials specialist AMSilk GmbH (Neuried, Germany) and biotech production specialst 21st.BIO (Copenhagen) have announced to join forces to...

