French Genkyotex SA has secured an €7.5m gross financing to expand the scope of its Phase II product GKT831 in Primary Biliary Cholangitis (PBC).
The company secured the proceeds through the issuance of convertible notes with share subscription warrants attached to YA II PN, LTD, an investment fund managed by Yorkville Advisors Global LP for up to €7.5m Upon signing, Genkyotex will receive €4.9m and controls the right to call an additional €2.45m from 80 to 95 days post transaction.
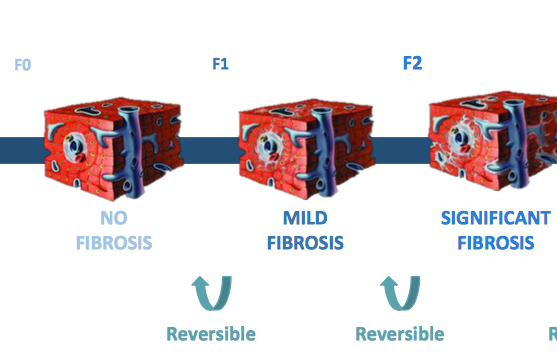
The company said, that the additional funding will support the design of an open label extension for an ongoing Phase II study with its anti-fibrotic compound GKT831 in the orphan liver disease Primary Biliary Cholangitis (PBC) - in which Genkyotex will finish an interim analysis this month. "Enrollment of our ongoing Phase II trial of GKT831 in Primary Biliary Cholangitis (PBC) has reached 86 patients, and to date no serious adverse events or liver-related adverse events have been reported," said Elias Papatheodorou, Chief Executive Officer of Genkyotex. Planning for the Phase 3 programme are underway. The money will also be used to support CMC activities for an NIH-funded Phase II trial of GKT831 in patients with idiopatic pulmonary fibrosis (IPF).
Genkyotex is listed on the Euronext Paris and Euronext Brussels markets. The company's pipeline consists of orally administered selective inhibitors of NOX enzymes. NOX enzymes 1-7 control multiple stress responses pathways simultaneously. Excessive stress responses end up in multiple diseases, particularly fibrosis, inflammation, pain processing, cancer, and neurodegeneration.
The lead product, GKT831, is a NOX1 (anti-angiogentic) and NOX4 (antifibrotic) inhibitor is currently clinically evaluated in a Phase II study in the orphan liver disease primary biliary cholangitis (PBC, a fibrotic orphan disease) and in an investigator-initiated Phase II study in Type 1 Diabetes and Kidney Disease (DKD). A grant from the United States National Institutes of Health (NIH) of $8.9 million was awarded to Professor Victor Thannickal at the University of Alabama at Birmingham (UAB) to fund a multi-year research programme evaluating the role of NOX enzymes in idiopathic pulmonary fibrosis (IPF), including a Phase II trial with the GKT831 in patients with IPF. This product candidate may also be active in other fibrotic indications such as NASH or scleroderma. The company's second anti-fibrotic product candidate, GKT771, is a NOX1 inhibitor targeting multiple pathways in angiogenesis, pain processing, and inflammation, currently undergoing preclinical testing.
Genkyotex also has a versatile platform well-suited to the development of various immunotherapies (Vaxiclase). A partnership covering the use of Vaxiclase as an antigen per se (GTL003) has been established with Serum Institute of India Private Ltd (Serum Institute), the world's largest producer of vaccine doses, for the development by Serum Institute of cellular multivalent combination vaccines against a variety of infectious diseases. This partnership could generate approximately €150m in revenues for Genkyotex, before royalties on sales.