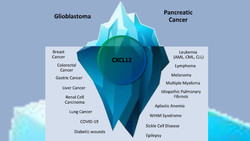
TME Pharma gets FDA Fast Track for Glioblastoma
TME Pharma N.V. with research sites in France and Germany (listed at Euronext Growth Paris: ALTME) is developing novel therapies for treatment of...

Ipsen puts US$1.8bn in pipeline expansion in movement disorders
Under the agreement, Skyhawk will provide its discovery platform for RNA-targeting small molecules across several therapeutic areas, including rare...

Curve Therapeutics raises £40.5m in Series A financing
New investors Columbus Venture Partners and British Patient Capital and founding investor Advent Life Sciences as well as seed investor Epidarex...

2:1 for new genomic techniques
At the end of December, genetic engineering sceptics celebrated that the Council of Agriculture Ministers narrowly failed to reach a qualified...

Pierre Fabre SAS in US$31m licence deal with Kinnate Biopharma Inc
With the US$31m agreement Pierre Fabre licences the global rights to exarafenib and other pan-RAF program assets from Kinnate...
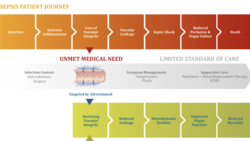
Shock: Adrenomed AG gets FDA fast track designation
After Adrenomed AG has demonstrated that its septic shock treatment led to a 60% reduction in relative 28-day mortality vs. placebo in a stratified...
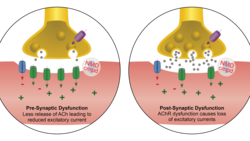
NDM Pharma provides POC for NMD670
In Science Translational Medicine the team headed by Thomas Holm Pedersen from NDM Pharma A/S report that they have conducted animal studies and a...

