The hunt for a cure for Huntington's
When Hans Meier knocks over a shelf for the second time in a row in just a few minutes, a colleague jests that he must have had too much to drink the night before. The 46-year-old German engineer tries to smile back, but decides not to reply. He doesn’t reveal what he has known for years – to be precise, since the morning of September 24, 2001. Meier had long suspected something was seriously wrong, but that was the day it was confirmed by the results of a genetic test. From that moment on, Meier knew with certainty that one day, things would begin to fall apart in earnest. That a leg would collapse beneath him, that an arm would begin to tick uncontrollably, that once familiar names would begin to escape him as his brain declined into dementia. He knew that years of long decay would follow. Years where he would slowly, but with cruel certainty, lose control over all of his physical and mental capacities. He knew that within 15 to 20 years of unstoppable and squalid decay, his disease would kill him. That fate is one all patients with the inherited neurodegenerative disorder Huntington’s disease (HD) continue to face – even though the underlying genetic mutation that causes it was uncovered back in 1993. Over a quarter of a century later, trials involving HD patients in the US and Europe are finally raising hopes the disease could be stopped, and that a cure might indeed be possible one day in the not all-too-distant future.
Early descriptions of the disease date back to the Middle Ages. The characteristic, uncontrollable movements sufferers often display lead to the name ‘chorea’, derived from the Greek word for ‘dance’. The discovery of what causes HD was one of the earliest successes in the then young research field of molecular genetics. And it was the personal success story of Nancy Wexler. HD had affected several members of Wexler’s immediate family, and desperate to find the cause, the neuropsychologist from Columbia University eventually pinpointed its source: a defective gene located on chromosome IV. The so-called huntingtin gene (HTT) has an unusual section made up of repeats of three of the four building blocks of DNA, abbreviated A, C, T, and G. In the gene, the number of repeating CAG sequences is slightly different in every individual. But in HD patients, this number hits a critical threshold of 35 repeats or higher. If a person has 36 or more CAGs in a row, the protein that is transcribed and translated from the gene turns toxic. It accumulates in the brain’s nerve cells over decades, and eventually kills the carrier.
A cure around the corner?
Wexler’s discovery made international headlines at the time. Suspected sufferers were soon able to check their genetic status, and find out whether they had inherited the mutation or not. But although the discoverer herself watched her mother die from the disease, she wasn’t keen to know about her own fate – one she couldn’t change. Without a therapeutic option, the scientist and many others have decided not to have the genetic test. “After finding the gene, I was totally disappointed that it took so long to actually do anything about it,” Wexler says today. She has since developed symptoms typical of HD.
For those who are only beginning to experience the onset of the disease, there is now a ray of hope. For the first time ever, a drug is being tested in HD that doesn’t treat only symptoms, but targets the underlying cause of the disease by attempting to disrupt the production of the toxic huntingtin protein. In mice and monkeys it proved able to capture the transcript copy (mRNA) of the gene and block its translation. Since August 2015, 46 patients in Canada, Britain and Germany have been treated with an ‘antisense oligonucleotide’ (ASO) developed by Californian biotech company Ionis Pharmaceuticals (formerly Isis).
Hans Meier is happy to be one of the guinea pigs. In August 2015, he received a call from his physician Patrick Weydt in the southern German city of Ulm – the centre of Europe’s Huntington’s network. About a third of the continent’s approximately 30,000 HD patients are registered and treated here. The neurologist, who now works at Bonn University Hospital, suggested that Meier take part in a Phase I trial with the Ionis test compound, and the engineer didn’t hesitate. Although he was still able to drive and communicate freely, and was in good physical condition, the signs of dawning HD were unmistakeable. He was still easily climbing the stairs to his apartment, although a bit stiff. But his arms stubbornly refused to remain at rest for more than a few seconds. And the urge to change his position when seated had grown very frequent.
Huntington’s – a family matter
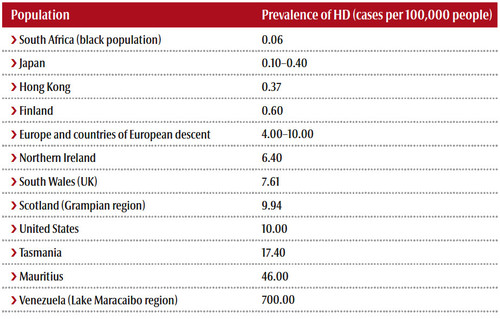 Although Meier’s mother also had the disease, it remained undiagnosed until shortly before her death. “She joined a sect, and we didn’t know whether the strangeness we observed in her behaviour was caused by that relationship or the symptoms of a disease.” She regularly forgot that her son was married, for example, or confused the past with the present. In 1998 she was finally diagnosed with Huntington’s. In 1999, she choked to death in a nursing home – a common complication for sufferers of HD as the swallowing reflex fades away.
Although Meier’s mother also had the disease, it remained undiagnosed until shortly before her death. “She joined a sect, and we didn’t know whether the strangeness we observed in her behaviour was caused by that relationship or the symptoms of a disease.” She regularly forgot that her son was married, for example, or confused the past with the present. In 1998 she was finally diagnosed with Huntington’s. In 1999, she choked to death in a nursing home – a common complication for sufferers of HD as the swallowing reflex fades away.
After that, it took her son two years to screw up enough courage to take the test. “Everyone forgets something occasionally, or gets a little confused. But after my mother’s diagnosis, I was always asking myself whether it was the first sign of HD,” Meier says. When the doctor at the Ulm clinic told him that he had indeed inherited the genetic predisposition for the condition, “it felt like someone had hit me in the face with a hammer.”
At that point, physicians couldn’t provide even a glimmer of hope to patients like Meier, even though theoretical approaches for blocking HD transcripts have been knocking around ever since the gene was discovered. And there’s a good reason why they’ve remained theoretical: the exact function of HTT in humans remains a mystery. Research has shown that it’s crucial for early development, because mouse embryos deprived of the gene die quickly. However, more recent work indicates it might not play an absolutely critical role in metabolism in adult animals. Another reason it’s taken decades to reach this point is that long-term studies in mice and monkeys had to be as thorough as possible before anyone was willing to take responsibility for a first test of Ionis’ AOS compound (HTT-Rx) in humans.
Safety before efficacy
A big hurdle for scientists has been a lack of technology able to differentiate between the two copies of HTT – the normal copy from one parent and the mutant copy from the other. “Both healthy and mutant RNA have CAG repeats,” says Weydt. It took scientists a long time to prove that targeting both wouldn’t harm a patient. What was important was to ensure that enough normal huntingtin protein remained in the cells. “The drug isn’t able to reduce amounts of huntingtin transcripts – either normal or mutant – below 50%,” says Weydt. Animal testing appears to show that it’s important to leave enough normal huntingtin protein, while at the same time inhibiting enough toxic huntingtin protein to show a therapeutic effect.
So far, so good. But every therapy comes with a downside. The Ulm trial‘s principal investigator Bernhard Landwehrmeyer explains that the downside in this case is that an antisense approach is based on RNA molecules that need to be tinkered with chemically to survive the attack of enzymes whose job it is to break down nucleic acids. “This means that for the body, the drug is a foreign entity,” he says. “And we can’t rule out that the immune system will jump on and reject such molecules, even though we didn’t observe such a reaction in animals.” That concern is certainly a valid one, as the potential drug would have to be administered every few weeks – probably lifelong. The primary goal of the Ionis Phase I trial, Landwehrmeyer stresses, is therefore not to prove efficacy, but to determine the safety of the compound. Regular MRI checks are mandatory in order to detect any signs of inflammatory response to antisense molecules.
More patients enrolled than originally planned
“Could you please stop wriggling,” a physician shouts at Meier through a microphone. It’s the patient’s second hour in a row in an MRI. “If I could stop wriggling, I wouldn’t be here,” Meier shouts back. This morning he received a first injection via a spinal infusion. Now the engineer needs to lie on his side and arch his back before being given a local anaesthesic and a spinal tap. After that, he’ll have to remain still for another hour as cerebrospinal fluid slowly trickles out. “And I’m not allowed to move the entire time,” Meier grumbles. The injection tops up the volume of lost cerebrospinal fluid. Afterwards, Meier has to walk around the clinic. The movement and diffusion should spread the injected fluid to his brain. A smoker, he’s allowed the occasional cigarette. During breaks from walking, Meier is required to perform many different types of tests: walking on tiptoe, walking on his heels, touching his nose, or answering endless questions on a number of forms. During other hourly breaks, the doctors take blood samples. “It’s excruciatingly boring,” Meier comments.
But he’s willing to endure it, even though he knows he might be in the control group, and receiving only a placebo. Or that even if he is being given the drug, it might not help. “I’m doing this for my kids,” the father of two says. He’s less worried about his own fate than theirs. It took him years to tell them what he discovered that day in 2001. Back then his children were 9 and 10. First he told himself that they were too young to understand. Then he decided to wait until they were 18. Then he wanted to let them finish studying first. Then he wanted them to enjoy just one more year of not knowing.
even though he knows he might be in the control group, and receiving only a placebo. Or that even if he is being given the drug, it might not help. “I’m doing this for my kids,” the father of two says. He’s less worried about his own fate than theirs. It took him years to tell them what he discovered that day in 2001. Back then his children were 9 and 10. First he told himself that they were too young to understand. Then he decided to wait until they were 18. Then he wanted to let them finish studying first. Then he wanted them to enjoy just one more year of not knowing.
Finally, his daughter called him while he was on a business trip in China to tell him she was pregnant. Instead of joy or pride, Meier felt a wave of panic, desperation and guilty grief. Why hadn’t he told her sooner about the family burden – the inherited mutation for HD that she might carry? The chances were 50%. Although terrified of their reaction, he finally broke the news that he had Huntington’s, and that the first symptoms had arrived. Now his children also have to live with the uncertainty, and the question of whether they should take a genetic test that could confirm a terrible future.
A cure – or even just a promising drug – could help ease the decision, simply by providing hope. The public won’t find out whether Ionis’ compound might be that drug before the end of 2017, and probably later. One sign of a possibly promising outcome is that the study has in the meantime enrolled more than the 36 patients originally planned. After the third of five infusions, Meier reports that his symptoms are fading. “And other than a light headache, I’ve suffered no side effects.”
Barriers to treatment
“The outcome regarding a recovery from or even reversion of HD symptoms depends on the areas of the brain the drug reaches – whether the brainstem, the cortex or the spinal cord,” Landwehrmeyer says. It’s still unclear whether there’s been sufficient penetration to the basal ganglia in the striatum, the brain component most affected by HD. Chances of improvement appear better when symptoms are less acute. And having enough nerve cells seems to be a prerequisite for the brain to recover and regenerate. “At least in HD mice treated with the antisense molecules, the symptoms cease on a behavioural, as well as a pathological level.” But the drug doesn’t work like aspirin. “Animal testing shows the antisense molecules take about 4-6 weeks to have an impact on disease symptoms,” Landwehrmeyer says. The effect lasts about four months. And fresh ASO has to be infused around every four weeks.
An RNAi solution?
Disadvantages inherent in ASO treatment could be overcome by a next generation of therapeutic concepts for HD. One drug that’s already in the pipeline is applied directly to the brain in a single dose. It was developed by Lisa Stanek, a former Harvard University scientist and the principle investigator at SanofiGenzyme’s labs in Massachussetts.
The company’s HD program grew out of a former collaboration with Ionis. The proof-of-concept, which with Ionis’ antisense molecules lowered huntingtin and essentially cured mice from HD, has since led to the development of a gene-therapy-based approach. Using adenoassociated viruses (AAV) as a transport vehicle, Stanek managed to deliver a message to the brain’s nerve cells that in effect instructed them to specifically destroy huntingtin gene transcripts. The message is encoded in an RNAi molecule – a single-stranded nucleic acid with a sequence that fits huntingtin transcripts. This activates the cell’s transcript shredder – the RNA interference mechanism. It’s able to sort one specific transcript out of a million and destroy it, while leaving all other transcripts untouched. “It can do the same thing as the Ionis drug,” says Stanek. “However, we use gene therapy to deliver it, meaning that we get essentially lifelong suppression of the huntingtin gene with just one dose.”
Stanek says Sanofi-Genzyme is developing the AAV/RNAi-based approach together with Boston-based biotech company Voyager. “We’ll file an IND next year, and hope to be in the clinic with it shortly after.” She has faith in her HD gene therapy approach because of her preclinical work with mice and monkeys. “Our HD mouse model recapitulates most of the features of the human disease,” Stanek says. The mice carry the entire human huntingtin gene with 128 CAG repeats. “That’s a lot, because we can’t wait around for years for these mice to get sick,” says Stanek. “We have to really push them to exhibit these disease phenotypes.” With 128 CAG repeats, the mice begin developing motor deficits at just three months.
Nevertheless, says the scientist, the therapy cleared the animals of HD symptoms. “But the mouse brain is a thousand times smaller than a human brain. The scale-up problem is enormous.” No injection in a single spot could reach all of the cells in the human brain. “That’s still science fiction at this point,” Stanek says. Luckily, it might not be necessary to treat the entire brain, as HD specifically affects two areas: the striatum and the cortex. And Stanek’s experiments in monkeys show that she can target those regions with current AAV vectors. “So we hope that when we scale-up to human patients, we can achieve the distribution necessary to translate that into a clinical benefit.”
Although AAV is known to be a safe vector, and has already been in brains of Parkinson’s patients for seven years without any adverse events, Stanek expects the US Food and Drug Administration to take an extremely cautious approach. “The FDA is going to put this under a microscope, because this is not a drug you can discontinue if patients do poorly. Once we put it there, the drug will be in their brains forever.” In terms of regulatory hurdles, Ionis’ ASO therapy is easier to develop, because if complications arise, the clinic could just stop infusions.
Trials – not too early, not too late
Stanek says if Genzyme’s AAV-RNAi trials go ahead, HD patients with early-stage symptoms will be eligible to take part. Although the drug should theoretically also help prevent the outbreak of HD in pre-symptomatic patients, it would be unethical to give them an experimental drug that could fail and harm them while they’re still healthy. On the other hand, “late-stage symptomatic HD patients won’t be eligible for this procedure (either), because their brains are already extremely deteriorated,” Stanek says.
Hans Meier is meanwhile eagerly awaiting the results of the Ionis trial. If the evaluation is positive, he’ll have the chance to continue the ASO therapy. Could it possibly stop his disease, or help his battered brain recover? Can he avoid his mother’s fate? Hope springs eternal, even though he’s already planned for the worst. “Doctor Weydt will get my body, so science can learn as much as possible,” Meier says. “After that, they’ll burn me. Everything’s going to be fine.”
(First published in European Biotechnology, Autumn Edition 2017)

