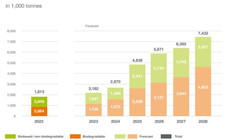
Bioplastics production capacity to quadruple by 2028
Global production capacity for bioplastics will almost quadruple within the next five years, according to estimates of the Nova Institute on behalf...
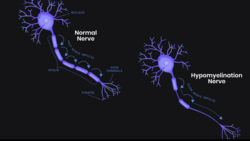
SynaptixBio Ltd gets FDA ODD for leukodystrophy
Using antisense oligonucleotides (ASO) to silence the beta tubulin 4a gene, Oxford-based SynaptixBio Ltd received its first ODD in early 2023 for a...
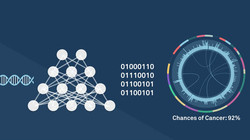
Roche leads Freenome Inc US$257m financing
Roche led the financing that will be used by San Francisco-based Freenome Inc to finance testing its early cancer detection platform in the Vallania...
Vidac Pharma Holding plc reports preclinical proof of principle
Vidac Pharma Holdings plc will report promising results for its cancer drug candidate VDA-1275 in multiple mouse cancer and human cellular...
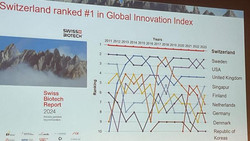
Swiss Biotech Report reporting record turnover
The Swiss Biotech Report 2024 commissioned by the Swiss Biotech Association and co-published with EY on the occasion of the Swiss Biotech Day...

AmphiStar BV secures €6m to expand production
The infusion of €6m in new capital from ECBF, Qbic III and FFTF will empower AmphiStar to kickstart commercial biosurfactant production in...

Nordic Bioproducts Group opens its first production facility
Nordic Bioproduct’s commercial-scale manufacturing site is expected to create €40m in revenue in the first full operating year. In...
