
AstraZeneca to acquire Amolyt Pharma for up to US$1.05bn
Under the agreement set to close in Q3/2024, AstraZeneca will acquire Amolyt Pharma for US$800m upfront and potential milestone payments of US$250m....
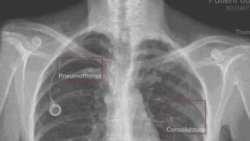
AZmed closes €15m Series A financing
AZmed said it will use the €15m to boost marketing of its proprietary artificial intelligence (AI) platform Rayvolve worldwide particularly in...

AMPLY Discovery Ltd bags £1.4m funding to tackle complex diseases
The 2021 spin-out from the Queen’s University Belfast announced it will close a seed financing round this year.vAMPLY is speeding up the discovery of...

New law to speed up clinical trials
The draft Medical Research Act (Medizinforschungsgesetz) presented by Health Minister Karl Lauterbach in Berlin before Easter is the centrepiece of...

Bayer SE secures rights to in €310m deal
Under the licence deal, BridgeBio Pharma, Inc. will pass the commercialisation rights for the transthyretin stabiliser acoramidis in Europe where...

2:1 for new genomic techniques
At the end of December, genetic engineering sceptics celebrated that the Council of Agriculture Ministers narrowly failed to reach a qualified...
Senisca Ltd raises £3.7m seed money
The proceeds of Senisca’s latest funding round led by Lifespan Vision Venture will support the preclinical development of the company’s...

