Innocent Meat bags €3m financing
The three-year-old startup announced it will use the new proceeds to optimise components of its automated plug-and-produce solution for mass...

Swiss vaccine developer Limma Tech get CARB-X grant
LimmaTech said, it will use the grant to advance preclinical development of its hexavalent vaccine candidate targeting Neisseria gonorrheae. Using...

Novo Nordisk to acquire Cardior Pharmaceuticals
The take-over agreement includes Cardior’s lead compound CDR132L, currently in Phase II clinical development for the treatment of heart failure....

Cambridge university spin-out HutanBio Ltd bags £2.25m seed financing
HutanBio, a Malaysian spin-out from scientists at Cambridge University, UK, has bagged a first tranche of seed investment from London-based Clean...
Relation Therapeutics Ltd bags US$35m in seed financing.
The latest financing round of generative AI drug discovery company Relation Therapeutics was led by DCVC and co-led by NVIDIA's venture arm,...
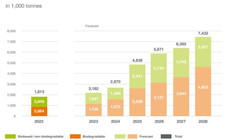
Bioplastics production capacity to quadruple by 2028
Global production capacity for bioplastics will almost quadruple within the next five years, according to estimates of the Nova Institute on behalf...
Forbion participates in US financing rounds
Forbion is throwing a few million into the ring in order to participate in oversubscribed financing rounds in the USA. Both Series B financings have...

