
Novo Nordisk to acquire Cardior Pharmaceuticals
The take-over agreement includes Cardior’s lead compound CDR132L, currently in Phase II clinical development for the treatment of heart failure....
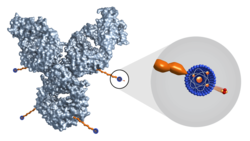
AstraZeneca to take over Fusion Therapeutics Inc
Fusion Pharmaceuticals Inc‘s acquisition for US$2bn upfront marks a major step forward in AstraZeneca delivering on its ambition to transform cancer...
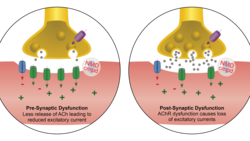
NDM Pharma provides POC for NMD670
In Science Translational Medicine the team headed by Thomas Holm Pedersen from NDM Pharma A/S report that they have conducted animal studies and a...
Lonza AG enters R&D collaboration with NeuroSense Therapeutics Ltd
Under the agreement with Lonza AG, NeuroSense Therapeutics Ltd will advance early diagnosis and treatment in the neurodegeneration field. Last...
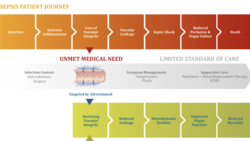
Shock: Adrenomed AG gets FDA fast track designation
After Adrenomed AG has demonstrated that its septic shock treatment led to a 60% reduction in relative 28-day mortality vs. placebo in a stratified...

Swiss vaccine developer Limma Tech get CARB-X grant
LimmaTech said, it will use the grant to advance preclinical development of its hexavalent vaccine candidate targeting Neisseria gonorrheae. Using...
Obesity: Arecor Therapeutics partners with TRx Biosciences
The collaboration of the biopharma and drug discovery specialists targets a potential US$80bn market of obesity drugs, according to Bloomberg...
